Hydrogen Plasma Smelting Reduction: Science, Technology, and the Path to Green Metals
The global metallurgical industry confronts an existential challenge of its own making: iron- and steelmaking alone account for approximately 7–9% of global CO₂ emissions, and the broader suite of primary metal production processes collectively constitute one of the largest anthropogenic sources of greenhouse gases on Earth. The replacement of carbon by hydrogen as the primary reductant constitutes the central strategic response to this challenge. Yet not all hydrogen-based routes are equivalent. Hydrogen Plasma Smelting Reduction (HPSR) represents a fundamentally distinct and uniquely powerful approach: a single-step, electrified, molten-state process in which the simultaneous melting and chemical reduction of mineral ores occurs within a thermal arc plasma, generating water vapour as the sole direct emission rather than CO₂. This review draws comprehensively on recent primary research to describe the underlying physics and thermodynamics, the phase transformation and microstructural kinetics, the extension of the method to non-ferrous feedstocks including nickel laterites and ilmenite, the emerging use of optical emission spectroscopy for in-situ process monitoring, and the strategic advantages of hybrid process architectures. Taken together, the evidence firmly establishes HPSR as one of the most promising technologies available to the sustainable metallurgy of the twenty-first century.
1. The Metallurgical Imperative: A Necessary Disruption
For over two centuries, iron and steel have been made by combusting carbon—in the form of coke or coal—both as the source of heat and as the chemical reductant that strips oxygen from iron ores. This chemistry is deeply embedded in the infrastructure of industrial civilisation, yet it is fundamentally incompatible with the climate targets set by the Paris Agreement, which demands a reduction of at least 80% in global CO₂ emissions by 2050. The scale of the problem is formidable: approximately 2.6 billion tonnes of iron ore are processed annually through the integrated blast furnace–basic oxygen furnace (BF-BOF) route, which contributes roughly 70% of world steel output and generates on average about 2.1 tonnes of CO₂ per tonne of crude steel. This single industrial process is, by itself, the largest individual industrial emitter of CO₂ on the planet. Iron- and steelmaking is thus responsible for approximately 6.5–7% of all anthropogenic CO₂ emissions.
The remaining 30% of steel is produced by melting scrap and directly reduced iron (DRI, or sponge iron) in electric arc furnaces (EAFs). While the EAF route yields 77% lower CO₂ emissions than conventional blast furnace steelmaking, it cannot alone address the scale of the problem, because the global demand for steel—projected to rise from the current approximately 1.8 billion tonnes per year toward 2.4 billion tonnes by 2040—will continue to require primary iron production from ores. Moreover, the quality and composition of post-consumer scrap is declining, placing additional constraints on scrap-based routes.
The use of hydrogen as a reductant is the principal candidate technology for decarbonising primary steelmaking. When hydrogen reduces iron oxide, the product is not CO₂ but water vapour: FexO+H2→xFe+H2OFexO+H2→xFe+H2O This simple reaction, if powered by renewably generated electricity and green hydrogen, yields no direct carbon emissions whatsoever. Hydrogen-based processes divide broadly into two families: (i) solid-state hydrogen direct reduction (DR) in shaft furnaces or fluidised-bed reactors, and (ii) hydrogen plasma smelting reduction (HPSR), in which the ore is simultaneously melted and reduced under a hydrogen-containing thermal plasma arc. These two approaches are not competing but potentially complementary, as shall be discussed in the context of hybrid process design below.
2. The Physics of Thermal Hydrogen Plasma
2.1 Plasma Generation and Species Composition
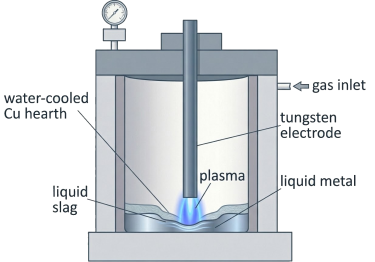
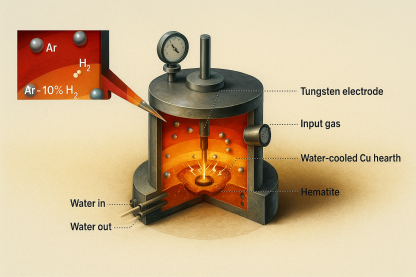
A thermal plasma is a partially or fully ionised gas at thermodynamic quasi-equilibrium, maintained by an electric arc. In HPSR, a direct-current (DC) or alternating-current (AC) arc is struck between a tungsten or graphite electrode and the input oxide charge, which simultaneously acts as the anode in DC configurations. The working gas is typically a mixture of argon and hydrogen—most commonly Ar-10%H₂—at pressures close to atmospheric. Argon serves as the plasma carrier gas, providing arc stability and the necessary electrical conductivity, while hydrogen provides the reducing species.
Within the arc plasma, the molecular hydrogen undergoes dissociation, excitation, and ionisation through mutual elastic and inelastic collisions between hydrogen particles and electrons. The resulting plasma contains a rich spectrum of hydrogen species: vibrationally ionised species (H⁺, H₂⁺, H₃⁺), excited atomic hydrogen (H*), and ground-state atomic hydrogen (H). Theoretical and experimental work has established that the reduction potential of these species decreases in the order H⁺ > H₂⁺ > H₃⁺ > H > H₂, meaning that the proton (H⁺) is by far the most potent reductant within the plasma. This hierarchy is of central importance: it means that a hydrogen plasma is not merely hot hydrogen gas, but a chemically qualitatively different reductant with drastically enhanced reducing power. As Bullard and Lynch demonstrated in early work, hydrogen in radical form possesses reducing power orders of magnitude greater than can be achieved with hydrogen gas alone.
The plasma temperatures generated in these arcs are extraordinary, ranging from approximately 4,500 K in the peripheral zones to 8,000–10,000 K or above in the arc core. Electron densities typically fall in the range of 10¹⁸ to 10²⁰ cm⁻³, values consistent with local thermal equilibrium (LTE) conditions that facilitate thermodynamic modelling of the arc zone. The degree of hydrogen dissociation within the arc—and hence the concentration of reactive radical species at the reaction interface—is governed by the competing ionisation and recombination events within the plasma volume and is directly sensitive to arc current, arc length, and gas composition.
2.2 Thermodynamic Advantages Over Molecular Hydrogen Reduction
The thermodynamic advantage of HPSR over solid-state direct reduction is profound and multi-faceted. In conventional molecular hydrogen reduction, the net reaction:
FexO+H2→xFe+H2OFexO+H2→xFe+H2O
has a positive (endothermic) net energy balance at the temperatures used in shaft furnaces (850–1000 °C), meaning that heat must be continuously supplied to drive the reaction forward. This
thermodynamic constraint imposes large energy penalties in industrial DR processes, where excess hydrogen must be circulated as a heat carrier.
In the plasma environment, by contrast, the reduction of wüstite (Fe_xO) by H⁺ ions at temperatures above 1600 °C is exothermic—the reaction releases heat, which is locally deposited at the reaction interface between the plasma and the molten oxide pool. This self-energising character is of considerable practical importance: the heat released by the plasma-driven reduction partially compensates for the electrical energy input, thereby improving the overall energy efficiency of the process. The equilibrium Gibbs free energy changes (ΔGΔG) for the reduction of iron oxide polymorphs by H⁺ ions become progressively more negative in the sequence wüstite < magnetite < hematite at temperatures around 1600 °C, confirming that all reduction steps are thermodynamically favourable under plasma conditions, even if wüstite reduction remains the kinetically most challenging step.
Furthermore, the presence of a molten oxide bath in HPSR eliminates several kinetic barriers that are intrinsic to solid-state DR. In the solid state, the formation of a dense iron product layer on the outer surface of the ore pellet progressively impedes the inward diffusion of H₂ and the outward diffusion of water vapour and oxygen, slowing the reduction rate dramatically once approximately 33% metallisation is achieved. In the molten state, by contrast, mass transport occurs by convection within the melt, driven by temperature gradients and electromagnetic stirring forces from the arc current, and the iron and oxide phases continuously segregate by density, exposing fresh unreduced oxide surfaces to the plasma.
Max Planck Institute for Sustainable Mat[...]
PDF-Dokument [3.1 MB]
3. Reaction Kinetics and Phase Transformation Sequence in Iron Ore Reduction
3.1 The Reduction Sequence
The reduction of hematite (Fe₂O₃) via HPSR follows the same formal sequence observed in conventional H₂-DR: Fe2O3→Fe3O4→FexO→FeFe2O3→Fe3O4→FexO→Fe However, the physical environment is radically different. In HPSR, these transformations occur in the molten state, with the entire oxide charge melted under the arc and the phase transformations occurring concurrently with reduction. Experimental work using interrupted Ar-10%H₂ plasma reduction of hematite pieces in a laboratory arc-melting furnace equipped with a tungsten electrode operated at 44 V and 800 A demonstrated that complete reduction to metallic iron can be achieved within approximately 15–30 minutes, depending on the mass of input material and the arc power.
During the initial reduction cycles, hematite is rapidly converted into a dual-phase magnetite-wüstite mixture. After 1 minute of plasma exposure, microstructural analysis by electron backscatter diffraction (EBSD) reveals a columnar grain structure with a strong solidification texture, consistent with rapid heat extraction to the water-cooled copper hearth. Phase maps confirm the coexistence of magnetite (weight fraction 0.24) and wüstite (weight fraction 0.76) after this first cycle. By 2 minutes, the sample is essentially fully wüstitic, with small iron domains of up to 10 µm becoming discernible, representing a metallic weight fraction of only 0.02. The bulk of the conversion from wüstite to iron occurs between 2 and 15 minutes, following a sigmoidal kinetic trend, with the maximum conversion rate of approximately 8 wt%/min reached at around 3.5 minutes.
3.2 The Rate-Limiting Step: Wüstite Reduction
In both H₂-based direct reduction and HPSR, the reduction of wüstite (Fe_xO) to metallic iron constitutes the rate-limiting step of the overall process. This step is thermodynamically the least favourable of the three sequential reduction reactions, particularly because the ΔGΔG for wüstite reduction by H⁺ ions at 1600 °C (approximately −2.9×10−3−2.9×10−3 kJ/mol) is less negative than those for magnetite and hematite reduction. As a consequence, the final 25% of the conversion (from 75% to 100% metallisation) requires the same duration as the first 75%, clearly reflecting the kinetic retardation imposed by wüstite reduction.
This phenomenon has a direct mechanistic explanation. The molten wüstite presents thermodynamically and kinetically unfavourable conditions for the nucleation of metallic iron within the oxide melt. Moreover, the interfacial region between iron and wüstite is progressively enriched in silicon (in the form of fayalite, Fe₂SiO₄), which was shown by atom probe tomography (APT) to segregate sharply to the wüstite/iron heterophase interface. This silicon enrichment creates an energetic barrier to further reduction at the interface. Importantly, however, as the HPSR process approaches completion, the plasma environment gradually removes these silicon-bearing impurities from the metal via sputtering and vapour-phase transport, so that the final iron product is almost entirely free of this contamination.
3.3 Kinetic Comparison with Solid-State Direct Reduction
A systematic comparison of HPSR kinetics with those of conventional H₂-based direct reduction demonstrates that the two methods yield broadly comparable reduction rates under optimised conditions, despite operating through entirely different mechanisms. In the solid state, complete reduction of iron ore pellets at 700 °C under a H₂ flow of 0.5 L/min requires approximately 100 minutes. At 850 °C under a flow of 2 L/min, complete reduction takes about 15 minutes, while at 1000 °C under 10 L/min it can be achieved in approximately 10 minutes. The HPSR process with Ar-10%H₂ achieves complete reduction in 15 minutes in an optimised laboratory configuration—comparable to the most aggressive solid-state DR conditions—but does so at a H₂ partial pressure of only 10% (versus 100% for the DR comparisons), and simultaneously produces liquid metal rather than solid sponge iron.
This comparison highlights a crucial practical advantage of HPSR: the process produces liquid iron in a single step, thereby bypassing the need for the energy-intensive re-melting of sponge iron in an EAF that is an obligatory post-processing step in the DR route. The transition from sponge iron through an EAF to liquid steel in current DR-based production typically consumes additional electrical energy of the order of 300–600 kWh per tonne, an energy penalty that HPSR entirely avoids.
Sustainable steel hydrogen plasma reduct[...]
PDF-Dokument [1.2 MB]
4. Microstructure and Chemistry of the Iron Product
4.1 Gangue Element Partitioning and Removal
One of the most remarkable features of HPSR is its remarkable self-cleaning character with respect to gangue-related impurities. High-resolution microstructural analysis combining EBSD, energy-dispersive X-ray spectroscopy (EDS), and atom probe tomography has provided atomic-scale insight into the fate of impurity elements throughout the reduction process. During the intermediate stages of reduction (5–15 minutes), gangue elements—in particular Si, Al, Ca, Mg, and Mn—partition into the remaining oxide regions and accumulate at the wüstite/iron interfaces, frequently forming fayalite (Fe₂SiO₄) structures in the interdendritic regions of the solidified wüstite.
APT analysis of the wüstite/iron interface in samples reduced for 5 minutes demonstrated that the reduced body-centred cubic (bcc) iron phase contains more than 98 at% iron, with all trace elements (Al, Ca, Mg, Mn, Si, Co, Ni, V) present at levels considerably lower than in the coexisting wüstite. A sharp enrichment of silicon is measurable at the very interface between the two phases, with the Si concentration rising over a distance of only a few nanometres—a finding that illuminates the atomic-scale mechanism by which silicon accumulates at the interface and impedes reduction. However, this enrichment is progressively eliminated as the reduction proceeds, with the plasma arc's sputtering action and vapour-phase transport mechanisms effectively volatilising these elements from the system.
By the time complete reduction is achieved, the resulting metallic iron is nearly free of harmful impurities: phosphorus and sulphur contents fall below 20 ppm (below the detection threshold of < 0.002 wt%), and silicon drops to approximately 30 ppm. This level of purity is directly competitive with high-quality commercially produced steel grades and does not require additional refining. The small residual fraction of oxide inclusions—worm-like structures containing Si and O, representing only 0.10 ± 0.05% of the sample volume—can be attributed to residual fayalite and does not compromise the quality of the product.
4.2 Carbon and Tramp Element Behaviour
A significant practical advantage of the HPSR process operating with an inert-gas/hydrogen mixture (Ar-10%H₂) and a non-carbonaceous electrode (tungsten) is the near-complete absence of carbon pick-up in the product iron. The carbon content of the final iron product in laboratory experiments was measured at approximately 60 ppm—only marginally higher than that of the starting hematite (approx. 60 ppm)—confirming that the process introduces no meaningful carbon contamination. This stands in stark contrast to the blast furnace route, which necessarily produces pig iron saturated in carbon (~4 wt%), requiring a subsequent oxygen-blowing step (the BOF) to reduce carbon content, with all the associated CO₂ emissions.
Copper and tungsten contamination from the hearth and electrode, respectively, were observed at low levels in partially reduced samples but dropped to 20 ppm (Cu) and negligible levels (W) in the fully reduced product, indicating that materials engineering of the reactor components can effectively control this issue at larger scales.
Nickel is a critical element in the shift to sustainable energy systems, with the demand for nickel projected to exceed 6 million tons annually by 2040'*,largely driven by the electrification of the transport sector. Primary nickel production uses acids and carbon-based reductants, emittingabout 20 tons of carbon dioxide per ton of nickel produced”,Here we present amethod using fossil-free hydrogen-plasma-based reduction to extract nickel from low-grade ore variants known as laterites. We bypass thetraditional multistep process and combine calcination, smelting, reduction and refining into a single metallurgical step conducted in one furnace. This approach produces high-grade ferronickel alloys at fast reduction kinetics. Thermodynamic control of the atmosphere of the furnace enables sel
Nature April 2025 Sustainable nickel ena[...]
PDF-Dokument [6.1 MB]
5. The Hybrid Strategy: Combining Direct Reduction and HPSR
5.1 Rationale and Hydrogen Efficiency
While HPSR offers compelling thermodynamic and kinetic advantages for the final stages of reduction and for producing liquid metal, it is not optimal across the entire reduction trajectory. This insight, developed in systematic efficiency studies, has led to the concept of a hybrid process that deliberately exploits the kinetically and energetically favourable regimes of both DR and HPSR.
The efficiency in H₂ utilisation—defined as the ratio of H₂ effectively consumed in reduction to the total H₂ supplied—differs markedly between the two processes as a function of metallisation degree. In solid-state DR, efficiency is highest at the beginning of the process (during the rapid conversion of hematite through magnetite to wüstite), where hydrogen has direct surface access to the reaction front. The efficiency then drops sharply at the onset of the wüstite reduction regime (approximately 33% reduction degree), because the thermodynamic conditions for complete wüstite reduction require a higher chemical potential of hydrogen, forcing a large excess to be supplied and recycled. Conversely, HPSR starts with unstable, inefficient behaviour in the early stages of reduction—when the arc tends to delocalize on cold, incompletely melted oxide—but becomes more stable and efficient once the ore has begun to liquefy and the reduction proceeds past approximately 33% metallisation.
These complementary efficiency profiles suggest an optimal crossover point, defined as the metallisation degree at which the H₂ efficiencies of DR and HPSR are equal. For DR conducted at 700 °C, this crossover occurs at approximately 38% reduction; at 900 °C, the crossover point moves to approximately 60% reduction. This temperature-dependence implies that lower DR temperatures, while intrinsically less energy-intensive for this step (since solid-state hydrogen reduction is endothermic), also permit an earlier transition to the plasma furnace, potentially reducing the overall reactor residence time and capital cost.
5.2 Experimental Validation of the Hybrid Route
Hybrid reduction experiments confirmed the expected advantages. Hematite pellets were partially reduced via DR at 700 °C to 38% metallisation (requiring approximately 10 minutes), then transferred to an Ar-10%H₂ plasma furnace for final conversion. The subsequent HPR step from 38% to full metallisation required only 10 minutes, compared to 15 minutes for a full HPR route starting from raw hematite—reflecting the improved arc stability when processing already partially metallised, electrically conductive material.
The total hydrogen consumption in the hybrid process was approximately 10.5 mol H₂ per mol of hematite, versus 22.7 mol H₂/mol hematite required to produce sponge iron via standalone DR at 700 °C. In other words, the hybrid route required six times less hydrogen in the DR step compared with a standalone DR route conducted to full metallisation. The total process time of the hybrid route (20 minutes) was only 5 minutes longer than a standalone HPR route (15 minutes), but achieved much better arc stability, substantially longer reactor lining lifetime, and considerably reduced hydrogen consumption.
The resulting iron product from the hybrid route shows a microstructure consistent with those described above for standalone HPSR, with a small volume fraction (approximately 0.01) of Fe- and Si-bearing oxide particles that float to the top of the melt pool as a slag-like layer and can be mechanically separated after solidification.
5.3 Industrial Implications and EAF Compatibility
A particularly compelling feature of HPSR and the hybrid route is their compatibility with existing EAF infrastructure. Electric arc furnaces already generate plasma discharges for the purpose of melting steel scrap, and much of the operational technology—arc control, magnetic stirring of the melt, gas injection systems, electrode and crucible design, water-based cooling, and protective slag formation—is directly transferable to HPSR reactors with only modest modifications. In a DC-operated EAF with a single cathode electrode and the furnace hearth as the anode, the configuration is directly analogous to the laboratory HPSR setups that have demonstrated the feasibility of the process. This means that the transition from the present industrial EAF fleet to hydrogen plasma furnaces does not require the construction of entirely new classes of plant, but rather incremental adaptation of existing assets—a critically important consideration for investment decisions in a capital-intensive industry facing urgent decarbonisation pressure.
Iron- and steelmaking cause ~7% of the global CO2 emissions, due to the use of carbon for the reduction of iron ores. Replacing carbon by hydrogen as the reductant offers a pathway to massively reduce these emissions. However, the production of hydrogen using renewable energy will remain as one of the bottlenecks at least during the next two decades, because making the gigantic annual crude steel production of 1.8 billion tons sustainable requires a minimum stoichiometric amount of ~97 million tons of green hydrogen per year. Another fundamental aspect to render the ironmaking sector more sustainable lies in an optimal utilization of green hydrogen and energy, thus reducing efforts for costly in-process hydrogen recycling. We therefore demonstrate here how the efficiency in hydrogen and en
2022 Green steel at its crossroads - Hyb[...]
PDF-Dokument [648.9 KB]
6. Extension to Non-Ferrous Feedstocks: Nickel Laterites
6.1 The Nickel Problem and HPSR as a Solution
Nickel presents a case study in sustainable metallurgy that closely parallels the situation for iron, but with still greater severity. Current primary nickel production generates approximately 20 tonnes of CO₂ per tonne of nickel produced—more than ten times the equivalent figure for steel. This staggering environmental footprint is a direct consequence of the energy- and carbon-intensive processing routes required, particularly for the dominant ore type: low-grade laterites, which account for 60% of all land-based nickel resources yet supply only about 40% of current annual production.
Laterite ores are subdivided into limonites and saprolites. In saprolites, nickel is not found as discrete minerals but is dissolved within complex magnesium-silicate frameworks—principally lizardite, (Mg,Fe,Ni)₃Si₂O₅(OH)₄, and pimelite, Ni₃Si₄O₁₀(OH)₂·4H₂O—making conventional beneficiation by froth flotation impractical. The standard industrial processing route for saprolites, the rotary kiln–electric furnace (RK-EF) process, involves ore drying in rotary dryers, calcination and partial pre-reduction in rotary kilns using coke, and final smelting in EAFs. This multi-step process accounts for approximately 87% of the total CO₂ emissions across the production chain, with the primary extraction step alone emitting 37.6–45.1 tonnes CO₂ per tonne of nickel via the RK-EF route. The energy demand is correspondingly immense, ranging from 230 to 570 GJ per tonne of nickel—far exceeding the 22 GJ per tonne required for steel.
6.2 HPSR of Nickel Laterites: A Single-Step Paradigm
Recent work demonstrated the feasibility of extracting metallic nickel from low-grade saprolitic laterite ores in a single metallurgical step using HPSR. This approach consolidates calcination, smelting, reduction, and refining into one process carried out within a single furnace charged with dried ore and an Ar–H₂ gas mixture, directly replacing the entire multi-stage RK-EF chain. The process operates on the principle that above approximately 1600 °C, the complex silicate minerals (lizardite, pyrope, pimelite) melt and dissociate into ionic species—Fe²⁺, Ni²⁺, Mg²⁺, O²⁻, and SiO₄⁴⁻. When the resulting ionic melt is exposed to hydrogen plasma, free oxygen (O²⁻) diffuses to the arc/melt interface and reacts with reactive hydrogen species to form water vapour, leaving behind two electrons per oxygen atom removed. These electrons are preferentially consumed by the metal cation with the lowest oxygen affinity—in this system, Ni²⁺—leading to the selective precipitation of metallic nickel ahead of iron.
This thermodynamically governed selectivity is of enormous practical significance. By controlling the stoichiometry of hydrogen introduced to the system (targeting an oxygen removal degree in the narrow interval of 20.7% to 21.3%), it is possible to produce a high-grade ferronickel alloy containing 70–90 wt% nickel, with corresponding Ni recovery rates of 60–90%. The resulting alloy is characterised by minimal impurities—less than 0.04 wt% Si, approximately 0.01 wt% P, and less than 0.09 wt% Ca—values considerably below those of commercial ferronickel alloys, which typically contain up to 0.3 wt% Si.
Phase evolution analysis by X-ray diffraction showed that the complex original ore minerals transform, under Ar plasma alone, into a simplified Mg-silicate system composed mainly of forsterite (Mg₂SiO₄, approximately 86 wt%) and pyroxenes (MgSiO₃, approximately 10 wt%). Progressive X-ray peak shifts toward higher 2θ angles with increasing hydrogen plasma exposure confirm a lattice contraction of the forsterite structure as Ni²⁺ and Fe²⁺ ions are progressively extracted from the crystal lattice, providing an elegant structural record of the reduction mechanism.
6.3 Process Flexibility and Atmosphere Control
Experiments with two different saprolitic ore compositions confirmed the versatility of the approach. Using a two-step reduction protocol with atmosphere control—first reducing in Ar-10%H₂ for 2 minutes, then completing the reduction in the leaner Ar-2.5%H₂ atmosphere to exploit the thermodynamic selectivity window—an overall ferronickel alloy grade of approximately 55 wt% Ni was achieved with a total Ni recovery of approximately 64%, at a cumulative processing time of only 4 minutes. Importantly, the process can, in practice, run continuously without stopping for gas adjustments, analogous to standard EAF operation.
From a life cycle perspective, preliminary calculations indicate that HPSR of nickel laterites has the potential to be approximately 18% more energy-efficient and to cut direct CO₂ emissions by up to 84% compared with the conventional RK-EF route. The Mg-silicate slags produced can be valorised as raw materials for brick, road, and cement production or potentially refined for cosmetic applications, adding further circularity to the process.
7. Extension to Waste Streams: Red Mud Valorisation
The processing of industrial waste streams constitutes another domain where HPSR demonstrates transformative potential. Bauxite residue, universally referred to as red mud, is the highly alkaline (pH 10–13) iron-rich waste generated by the Bayer process for alumina extraction. With a global stockpile exceeding 4 billion tonnes and annual production of approximately 120–150 million tonnes, red mud represents one of the largest and most hazardous industrial solid waste streams on Earth. Its strongly alkaline character and high heavy metal content render it dangerous to ecosystems and difficult to neutralise or repurpose.
Work by Jovičević-Klug and colleagues demonstrated that HPSR can extract high-purity metallic iron from red mud in a single melting step, simultaneously neutralising the alkaline character of the residual slag. Crucially, the plasma environment does not merely reduce the iron oxides; it also transforms the mineralogy and chemistry of the resulting slag such that the residue acquires a near-neutral pH of approximately 7.5 and a glassy, silicate-dominated structure. This transformation from a toxic, caustic waste material into a metallurgically valuable product (high-purity iron) and a construction-industry-compatible slag represents a paradigmatic application of circular metallurgy principles. The final metallic iron produced from red mud via HPSR achieves a purity of approximately 98 wt%, with the residual oxides exhibiting neutral pH—significantly reducing the costs and environmental burdens associated with conventional red mud neutralisation treatment.
8. Ilmenite: Exploiting HPSR for Titanium and Iron Co-Production
8.1 The Industrial Context
Ilmenite (FeTiO₃) occupies a position of strategic importance in both the titanium and steel industries. It supplies approximately 90% of global demand for titanium minerals, with world production reaching approximately 8,900 thousand tonnes in 2024 and reserves exceeding 510,000 thousand tonnes. The conventional processing of ilmenite—sulphate process, chloride process, or carbothermic EAF smelting—relies on acid digestion and carbon-based reductants, generating substantial waste streams and CO₂ emissions.
HPSR offers a fundamentally different approach to ilmenite processing, exploiting the well-known thermodynamic stability hierarchy between iron oxides and titanium oxides under hydrogen plasma conditions. The reduction potential of the Ar-10%H₂ plasma at the temperatures achieved in laboratory reactors (up to approximately 8,000–9,000 K in the arc core) is sufficient to reduce iron oxide to metallic iron, while TiO₂ remains stable in the slag—achieving effective metal/slag separation in a single molten-state step.
8.2 Experimental Results and OES Monitoring
Experiments with industrial ilmenite (46.8 wt% TiO₂, 39.4 wt% FeO, 8.4 wt% Fe₂O₃) under Ar-10%H₂ plasma for durations of 150–750 seconds demonstrated progressive iron recovery, reaching approximately 70% metallisation at the longest reduction time. The metallic droplets produced at 600 seconds contained 92.31 wt% Fe, with oxygen and silicon reduced to 6.84 wt% and 0.37 wt% respectively, and no titanium oxide was detected in the metal phase—confirming its complete retention in the slag. This slag, enriched in TiO₂, represents a valuable intermediate for downstream titanium extraction, potentially achieving the desired separation in a single furnace operation without acid digestion.
These experiments were monitored in real time by optical emission spectroscopy (OES), enabling unprecedented insight into the temporal evolution of the reduction process. The characteristic emission lines of H I (at 486 nm, Hβ), Fe I (dominant in the 350–500 nm range), Ti I (approximately 500 nm), and gangue element lines (Mg I, Mn I, Ca II, Al I) were identified and tracked throughout the experiments. The progressive consumption of hydrogen was revealed by the gradual decline in Hβ intensity after an initial peak, while the increasing Fe I intensity marked the progressive formation of metallic iron. The Ti I intensity showed a characteristic rise and then decline, reflecting the evolving slag surface coverage as iron oxides were progressively depleted and titanium oxide became the dominant phase at the slag surface.
9. In-Situ Process Monitoring by Optical Emission Spectroscopy
9.1 The Challenge of HPSR Process Control
The highly dynamic, non-linear behaviour of thermal plasma arcs—and their complex interaction with the evolving molten oxide bath—constitutes one of the principal technical challenges for the scale-up and industrial implementation of HPSR. Real-time knowledge of the plasma composition, temperature, electron density, and reduction progress is essential for process control and optimisation, yet the extreme temperatures (exceeding 5,000–10,000 K) and the aggressive chemical environment preclude the use of conventional immersion-based analytical methods.
9.2 OES as a Non-Intrusive Diagnostic Tool
Optical emission spectroscopy has emerged as the pre-eminent technique for non-intrusive, in-situ monitoring of HPSR reactors across scales ranging from 15-gram laboratory arc-melting furnaces to 9,500-gram pilot-scale reactors. OES operates by collecting the light emitted by the plasma through an optical fibre and quartz viewport and resolving it spectrometrically across the wavelength range of approximately 200–700 nm, where characteristic emission lines of all relevant species—hydrogen, iron, and the principal gangue elements—are found.
The plasma temperature is calculated from the Boltzmann equation applied to multiple Fe I optical emission lines across different energy levels and electron densities are derived from the Stark broadening of the hydrogen Balmer Hβ line (486 nm) using established empirical models. Plasma temperatures in the range of 4,500–9,000 K and electron densities between 10¹⁸ and 10²⁰ cm⁻³ have been measured at laboratory scale, consistent with values predicted by LTE models.
9.3 Key Process Indicators Accessible by OES
In systematic studies across three reactors of different scales—at the Max-Planck-Institut für Eisenforschung (MPIE), Montanuniversität Leoben (MUL), and K1-MET GmbH—OES was shown to provide several distinct categories of process-relevant information.
The Hβ/Ar I intensity ratio tracks hydrogen consumption: a decreasing ratio indicates that hydrogen is being consumed in reduction reactions. Conversely, the reappearance of strong hydrogen lines in a previously iron-dominated spectrum signals that the reduction intensity has decreased, potentially due to completion of reduction or to exhaustion of the available oxide. The Fe I/Ar I and FeO/Ar I ratios track the formation and evaporation of metallic iron and the persistence of liquid oxide, respectively; increasing FeO molecular emission (a broadband structure from 550 to 650 nm with maxima at approximately 565, 590, and 625 nm) during the early stages of reduction signals the presence of iron oxide in the melt, while its subsequent disappearance tracks the progress of metallisation. The emergence of strong gangue element lines (Ca II, CaO, Mg I, Na I) in the later stages of reduction signals the formation of a slag layer on top of the melt, as the increasingly metal-depleted slag begins to contribute its volatile and excited species to the plasma.
A particularly instructive finding from pilot-scale experiments at K1-MET was the characteristic spectral signature of iron ore fines entering the plasma zone during continuous charging: the arrival of ore was immediately reflected in a dramatic broadband increase in FeO molecular emission and Fe I atomic emission, providing a direct real-time indicator of the charging rate and the onset of reduction in the newly added material. This level of process observability is an essential prerequisite for the closed-loop control systems that will be required for industrial HPSR operations.
10. Advantages of HPSR: A Synthesis
The scientific and technological evidence reviewed above collectively identifies HPSR as a technology of exceptional promise for sustainable metallurgy. Its principal advantages over both conventional carbon-based reduction and over purely molecular hydrogen-based DR may be summarised as follows.
Single-step liquid metal production. HPSR produces liquid iron directly from ore, without the intermediate step of producing solid sponge iron that must subsequently be melted in a conventional EAF. This circumvents a significant energy penalty and simplifies the process chain.
Low hydrogen consumption. Despite achieving rapid and complete reduction, HPSR requires remarkably lean hydrogen gas mixtures—as little as 10% H₂ in the working gas—sufficient to achieve full metallisation. This is because the plasma converts even a small quantity of molecular H₂ into highly reactive radical species with dramatically enhanced reducing power.
Compatibility with complex and low-grade feedstocks. Solid-state DR requires high-grade, pre-processed pellets of relatively uniform composition. HPSR, by contrast, processes the entire ore charge in the molten state, disaggregating the mineralogical complexity of low-grade feedstocks—including saprolitic nickel laterites, ilmenite concentrates, and industrial waste streams such as red mud—into simple ionic species that react selectively with the plasma.
Self-cleaning chemistry. The high-temperature plasma environment, combined with the volatility-driven removal of impurity elements and the density-driven segregation of slag from metal, produces metallic products of very high purity. Harmful impurities (P, S) fall below 20 ppm, and gangue elements (Si, Al, Ca) are effectively removed without additional refining steps.
Selective reduction for tailored alloy composition. By thermodynamic control of the H₂ partial pressure and the duration of plasma exposure, it is possible to achieve selective reduction of specific metal cations—exploiting their differing affinities for oxygen—to produce tailored alloy compositions. This has been demonstrated for nickel laterites, where high-grade ferronickel with up to 90 wt% Ni can be obtained by careful atmospheric control.
Waste valorisation and circular economy integration. HPSR treats industrial waste streams (red mud, zinc-bearing slags) as valuable feedstocks rather than as disposal problems. The resulting slags, detoxified and compositionally transformed by the plasma environment, find ready application in construction materials, closing material cycles and eliminating the environmental liability of indefinite waste stockpiling.
EAF infrastructure compatibility. The technical knowledge and hardware base of the existing global EAF fleet—arc control, electrode technology, refractories, stirring, gas injection—is directly and largely transferable to HPSR furnaces, minimising the capital requirements and technology risk for the transition.
CO₂ emission reduction potential. The combination of zero direct carbon emissions (water vapour is the only by-product of hydrogen reduction), high energy efficiency (from the self-energising character of the exothermic wüstite reduction under plasma conditions), and the elimination of multiple processing steps yields dramatic reductions in specific CO₂ emissions. For nickel, modelling suggests up to 84% reduction versus the RK-EF route; for iron, comparison with the BF-BOF route implies elimination of the approximately 2.1 t CO₂/t steel directly attributable to the carbon-based reduction chemistry.
11. Remaining Challenges and Outlook
Despite the compelling evidence outlined above, several challenges must be addressed before HPSR can be realised at industrial scale. The most fundamental constraint is the availability and cost of green hydrogen: making the global annual steel output of approximately 1.8 billion tonnes sustainable by purely hydrogen-based methods requires a minimum stoichiometric consumption of approximately 97 million tonnes of green hydrogen per year, far exceeding current global green hydrogen production capacity. The hybrid process strategy—using solid-state DR to reduce ore to the crossover point, then completing the conversion in HPSR—substantially alleviates this constraint by maximising hydrogen utilisation efficiency.
Arc stability in the early stages of HPSR, particularly when processing raw (non-conductive) oxide ores, remains a technical challenge. The arc tends to delocalize and scatter on incompletely melted material, reducing efficiency and potentially damaging electrodes and refractory linings. This issue is substantially mitigated in the hybrid route (where partially metallised, conductive material is charged) and can be addressed at scale through optimised electrode design, slag engineering, and the application of existing EAF arc-stabilisation technologies.
Process monitoring and control—the subject of ongoing OES development—must be advanced to the level of full closed-loop automation before industrial deployment becomes viable. The relationship between OES spectral signatures and the key process variables (reduction degree, melt composition, hydrogen consumption, electrode wear) must be fully quantified across the range of reactor scales and ore types that will be encountered in industrial practice. The work reviewed here represents the foundational experimental and analytical basis for this quantification.
Finally, the scale-up trajectory from laboratory reactors (15–30 grams of ore) through pilot scale (K1-MET, up to 90 kg) to industrial scale (hundreds of tonnes per heat) presents well-known challenges in plasma engineering: arc stability at high currents, mass transport in large molten baths, refractory lifetime, and electrode consumption all require sustained engineering development. Promising technological solutions—multi-electrode AC configurations, external electromagnetic stirring, bottom gas injection for melt homogenisation—are all established in the EAF industry and can be systematically adapted for HPSR.
12. Conclusions
Hydrogen Plasma Smelting Reduction stands at a remarkable convergence of scientific depth and technological urgency. The fundamental thermodynamic and kinetic advantages of operating in the plasma state—exothermic wüstite reduction under H⁺ bombardment, elimination of solid-state mass-transport barriers, simultaneous smelting and reduction, and self-cleaning impurity removal—are now documented with atomic-scale precision using state-of-the-art characterisation tools including atom probe tomography, high-resolution EBSD, and in-situ optical emission spectroscopy. These advantages translate directly into reduced CO₂ emissions, higher product purity, greater feedstock flexibility, and lower capital costs relative to conventional pyrometallurgical routes.
The extension of HPSR beyond iron ore to nickel laterites, ilmenite, and hazardous industrial waste streams such as red mud demonstrates that the technology is not a narrowly specialised niche process but a general platform for sustainable primary metal production. As the global energy system decarbonises and green hydrogen becomes progressively more available and economically competitive, HPSR—particularly in hybrid combination with solid-state direct reduction—offers a technically mature and industrially credible pathway to the carbon-neutral metallurgy that the twenty-first century demands.
References
- Souza Filho, I. R., Ma, Y., Kulse, M., Ponge, D., Gault, B., Springer, H., & Raabe, D. (2021). Sustainable steel through hydrogen plasma reduction of iron ore: Process, kinetics, microstructure, chemistry. Acta Materialia, 213, 116971. https://doi.org/10.1016/j.actamat.2021.116971
- Souza Filho, I. R., Springer, H., Ma, Y., Mahajan, A., da Silva, C. C., Kulse, M., & Raabe, D. (2022). Green steel at its crossroads: Hybrid hydrogen-based reduction of iron ores. Journal of Cleaner Production, 340, 130805. https://doi.org/10.1016/j.jclepro.2022.130805
- Manzoor, U., Mujica Roncery, L., Raabe, D., & Souza Filho, I. R. (2025). Sustainable nickel enabled by hydrogen-based reduction. Nature. https://doi.org/10.1038/s41586-025-08901-7
- Pauna, H., Ernst, D., Zarl, M., Souza Filho, I. R., Kulse, M., Büyükuslu, Ö., Jovičević-Klug, M., Springer, H., Huttula, M., Schenk, J., Fabritius, T., & Raabe, D. (2024). The Optical Spectra of Hydrogen Plasma Smelting Reduction of Iron Ore: Application and Requirements. Steel Research International, 2400028. https://doi.org/10.1002/srin.202400028
- Sun, Y., Pauna, H., Torres Mejia, L. G., Klapproth, D., Manzoor, U., Souza Filho, I. R., Tanskanen, P., Fabritius, T., & Raabe, D. (2025). Monitoring Hydrogen Plasma Smelting Reduction of Ilmenite by Optical Emission Spectroscopy at Laboratory Scale. Journal of Sustainable Metallurgy. https://doi.org/10.1007/s40831-025-01200-5
- Jovičević-Klug, M., Souza Filho, I. R., Springer, H., Adam, C., & Raabe, D. (2024). Green steel from red mud through climate-neutral hydrogen plasma reduction. Nature, 625, 703–709.
- Raabe, D. (2023). The materials science behind sustainable metals and alloys. Chemical Reviews, 123, 2436–2608.