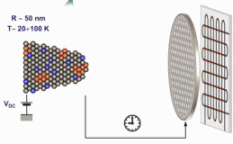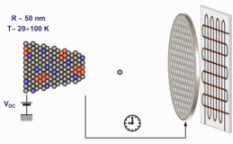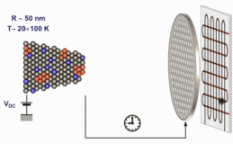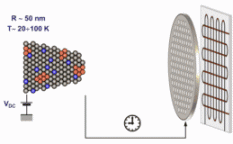
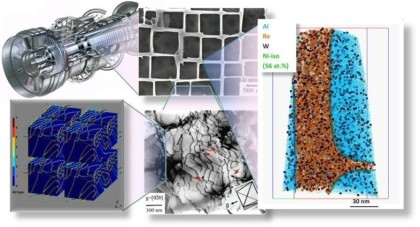
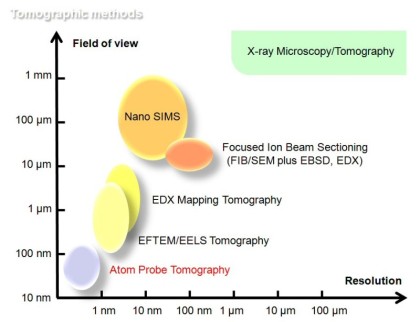
Motivation for using tomographic atom probe analysis
Richard Feynman said that if he were to express his scientific life experience in one sentence he would state that 'Matter is made of Atoms' much as already rendered by Demokrit 2500 years before him.
Yet, seeing and mapping atoms in complex materials and revealing the associated structure - composition - property relationships at this very scale remains a grand challenge in modern materials
science, physics and chemistry.
The properties of materials emerge from very short-ranged interactions among the atoms that they are composed of and their environment. Therefore, we need to retrieve as much correlated information
as possible about the structure, properties and dynamic behavior of materials at the atomic and electronic scales, and relate this to the associated properties in order to develop better materials
for advanced engineering and functional applications. Only with this close atomic scale tie between structure and properties can we work out the minutiae of materials processes and tailor
improvements of advanced, next generation materials.
Despite the already impressive current performance of state-of-the-art analytical techniques, we are still far from reaching the ultimate resolution limit, i.e. obtaining all relevant information
required for the full understanding of a real material at the atomic and electronic scales in 3D.
What is Atom Probe Tomography
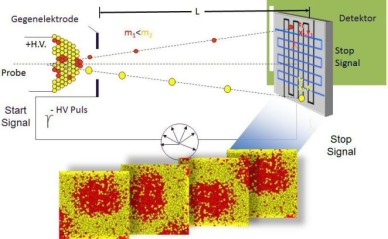
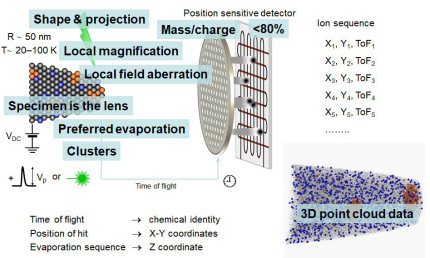
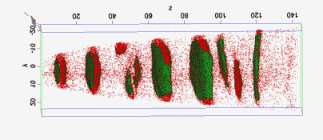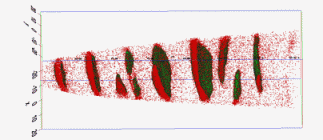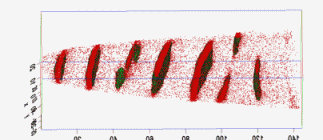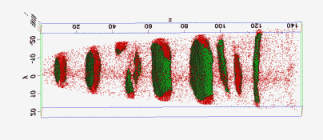
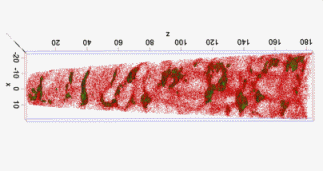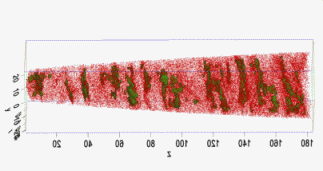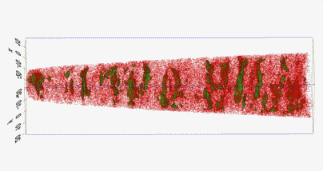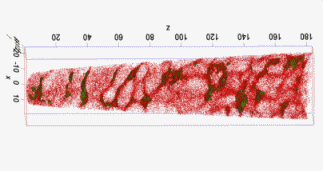
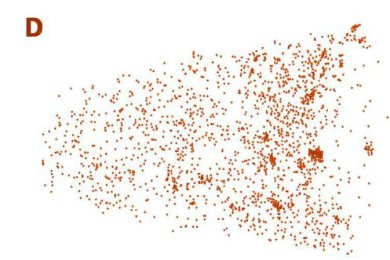
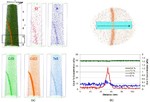
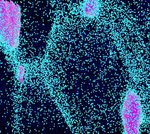
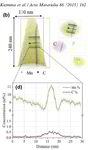
The figure above illustrates the principles of APT. By applying either ultra-fast high voltage or laser pulses to a positively biased tip-shaped specimen (typically having a radius of curvature ~100 nm), surface atoms are field-evaporated layer by layer. The field-evaporated atoms are ionized and accelerated towards a position sensitive detector that records their time of flight (for chemical analysis) and impact positions (for determination of the original position within the sample). A three-dimensional elemental map can be reconstructed from the collected data, applying an inverse projection of the detector coordinates to the tip surface. Depth-scaling is done taking into account the sequence and volumes of detected atoms. Typical dimensions of the analyzed volume are 100 × 100 × 200 nm3 for state-of-the-art instruments.
The APT method is based on the controlled field evaporation of surface atoms from a tip-shaped sample, which can be achieved by applying a combination of a high electrical field and either: (1) ultra-fast voltage pulses or (2) ultra-fast laser pulses. The second mode of operation requires the purchase also of an optional laser pulse module, which we plan for our investment. The laser module is essential for analyzing materials of low electrical conductivity and/or high brittleness and hence tremendously widens the range of materials that can be studied by APT.
One LEAP 3000 atom probe system and two Leap 5000 atom probe systems are operative at the Max-Planck-Institut für Eisenforschung (MPIE).
Ultrastrong steel via minimal lattice mi[...]
PDF-Dokument [6.2 MB]
M. Kuzmina, M. Herbig, D. Ponge, S. Sandlöbes, D. Raabe
Science 349, 1080 (2015)
Linear complexions: Confined chemical and structural states at dislocations
Dislocations Linear Complexions Science [...]
PDF-Dokument [1.6 MB]
For 5000 years, metals have been mankind’s most essential materials owing to their ductility and strength. Linear defects called dislocations carry atomic shear steps, enabling their
formability. We report chemical and structural states confined at dislocations. In a body-centered cubic Fe–9 atomic percent Mn alloy, we found Mn segregation at dislocation cores during
heating, followed by formation of face-centered cubic regions but no further growth. The regions are in equilibrium with the matrix and remain confined to the dislocation cores with
coherent interfaces.The phenomenon resembles interface-stabilized structural states called complexions. A cubic meter of strained alloy contains up to a
light year of dislocation length, suggesting that linear complexions could provide
opportunities to nanostructure alloys via segregation and confined structural states.
Atomic-Scale Quantification of Grain Boundary Segregation in Nanocrystalline Material
M. Herbig, D. Raabe,Y. J. Li, P. Choi, S. Zaefferer, and S. Goto ,2PRL 112, 126103 (2014) PHYSICAL REVIEW LETTERS
Phys Rev Lett. 2014 grain boundary segre[...]
PDF-Dokument [842.8 KB]
Grain boundary segregation leads to nanoscale chemical variations that can alter a material's performance by orders of magnitude (e.g., embrittlement). To understand this phenomenon, a large number of grain boundaries must be characterized in terms of both their five crystallographic interface parameters and their atomic-scale chemical composition. We demonstrate how this can be achieved using an approach that combines the accuracy of structural characterization in transmission electron microscopy with the 3D chemical sensitivity of atom probe tomography.
Thsi approach is referred to as correlative atom probe tomography.
Herer, we find a linear trend between carbon segregation and the misorientation angle ω for low-angle grain boundaries in ferrite, which indicates that ω is the most influential crystallographic parameter in this regime. However, there are significant deviations from this linear trend indicating an additional strong influence of other crystallographic parameters (grain boundary plane, rotation axis). For high-angle grain boundaries, no general trend between carbon excess and ω is observed; i.e., the grain boundary plane and rotation axis have an even higher influence on the segregation behavior in this regime. Slight deviations from special grain boundary configurations are shown to lead to unexpectedly high levels of segregation.
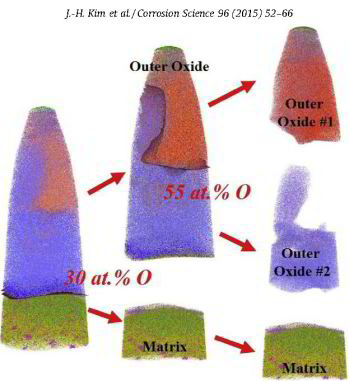
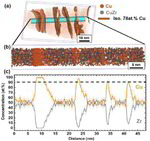
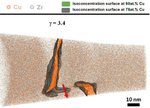
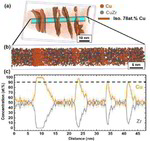
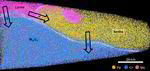
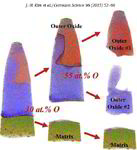
The role of grain boundaries in the initial oxidation behavior of austenitic stainless steel containing alloyed Cu at 700 °C for advanced thermal power plant applications
Corrosion Science 96 (2015) 52-66
Ju-Heon Kim, Byung Kyu Kim, Dong-Ik Kim, Pyuck-Pa Choi, Dierk Raabe, Kyung-Woo Yi
Corrosion Science 96 (2015) 52 Atom prob[...]
PDF-Dokument [2.0 MB]
Despite enormous research efforts into renewable energy technologies, conventional thermal power plants are still by far the most important power generation source worldwide;
thermal power plants are expected to maintain this position for at least several decades. All power generation sources based on fossil fuel burning inevitably produce CO2, one of the
major causes of global warming. Considering the contribution to the total CO2 emission, improving fossil fuel-based energy conversion efficiency to 60% by increasing the reaction
temperature is the most efficient way to reduce total CO2 emissions by up to 30%. Ultra supercritical (USC) coal-fired thermal power plants are operated at a steam pressure of P24.1
MPa and a steam temperature of P593 °C [4,5] in the boiler tube main line, such as in the superheater and reheater. These conditions require new boiler tube materials that can resist these
extreme temperature and pressure conditions. In this context, there are two main requirements for boiler tube materials: creep strength and oxidation resistance. Austenitic
stainless steels are considered candidate alloys for boiler tubes due to their higher strength at these operating conditions compared with the previously used ferritic stainless
steels. Austenitic stainless steels containing alloyed Cu were developed in the 1990s based on conventional 304 stainless steels, by the
addition of Cu to form stable copper precipitates. Similar lattice parameters and a small interfacial energy between the copper precipitates and austenitic matrix retard precipitate growth
at high temperature, enabling the material to maintain its high creep strength. Steam-side oxidation of the boiler tube is one of the main
mechanisms of power plant failure aside from creep rupture, where the failure occurs due to poor adhesion of the oxide. In particular, Fe-rich oxides are considered poorly adhesive and
non-protective, rendering the alloy susceptible to cracks and spallation during operation. The lifetime of boiler tube materials depends strongly on their resistance to oxidation.
Here, the role of grain boundaries during the early stages of oxidation in austenitic stainless steels containing alloyed Cu was investigated using APT, TEM, EBSD, EPMA, and XRD. The oxidation experiments were performed at 700 °C in air with 20% water vapor. Within 4 lm from the grain boundaries, the oxide layer exhibits a dual-layer structure consisting of a thin Fe-rich spinel oxide on a protective Cr2O3 oxide. Away from the grain boundaries, non-protective spinel oxide layers are formed as the outer and inner oxide layers. A critical grain size that prevents the formation of fast-growing spinel oxides is discussed.
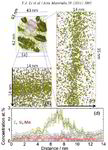
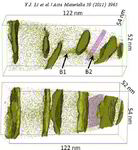
Atom probe informed simulations of dislocation–precipitate interactions reveal the importance of local interface curvature
A. Prakash, J. Guenole, J. Wang, J. Mü ller, E. Spiecker, M.J. Mills, I. Povstugar, P. Choi, D. Raabe and E. Bitzek
Acta Materialia 92 (2015) 33 Atom probe [...]
PDF-Dokument [1.8 MB]
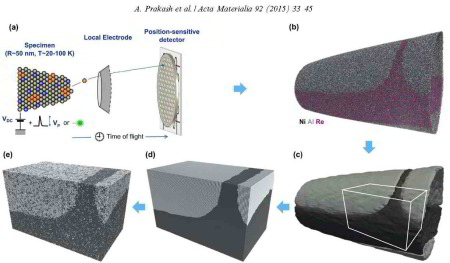
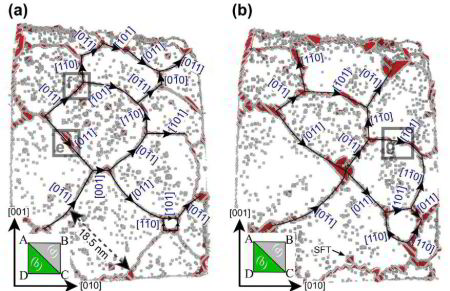
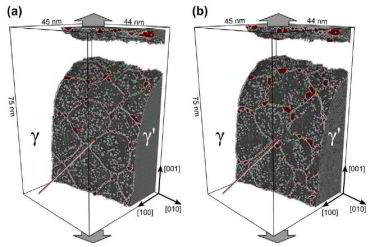
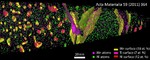
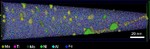
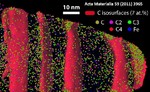
The interaction of dislocations with precipitates is an essential strengthening mechanism in metals, as exemplified by the superior high-temperature strength of Ni-base superalloys. Here we use atomistic simulation samples generated from atom probe tomography data of a single crystal superalloy to study the interactions of matrix dislocations with a gamma' precipitate in molecular dynamics simulations. It is shown that the precipitate morphology, in particular its local curvature, and the local chemical composition significantly alter both, the misfit dislocation network which forms at the precipitate interface, and the core structure of the misfit dislocations. Simulated tensile tests reveal the atomic scale details of many experimentally observed dislocation–precipitate interaction mechanisms, which cannot be reproduced by idealized simulation setups with planar interfaces. We thus demonstrate the need to include interface curvature in the study of semicoherent precipitates and introduce as an enabling method atom probe tomography-informed atomistic simulations.
Acta Materialia 86 (2015) 34
Acta Materialia 86 (2015) 34 Dynamic str[...]
PDF-Dokument [696.9 KB]
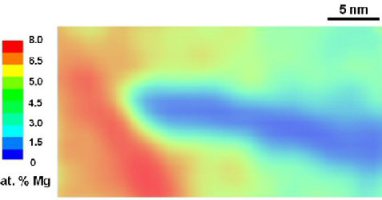
Dynamic strain aging arises from the interaction between solute atoms and matrix dislocations in strained metallic alloy. It initiates jerky dislocation motion and abrupt softening, causing
negative strain rate sensitivity. This effect leads to instable flow phenomena at the macroscopic scale, appearing as a serrated stress–strain response and deformation banding. These macroscopic
features are referred to as the Portevin–Le Chatelier effect (PLC). Here we study the atomistic origin of dynamic strain aging in an Al-4.8 at.% Mg alloy using atom probe tomography (APT)
and transmission electron microscopy (TEM). Samples were prepared from as-cold rolled (90% thickness reduction), stabilized (120°C, 20 h) and recrystallized sheets (400°C, 10 min),
respectively. In the stabilized state, Mg was found to decorate <110> aligned dislocations with up to 12.5 at.%. Tensile tests in combination with thermographic and laser speckle
observations were used to map the deformation bands for the site-specific extraction of APT samples from regions inside the PLC bands. We observed an asymmetrical Mg distribution along some of
the dislocations, matching model predictions for high dislocation speeds at peak drag stress by Zhang and Curtin. In this case, the Mg distribution is characterized by depletion
in the compressive regime above the dislocation slip plane and enrichment in the dilatation region below the slip plane. Mg also depletes in a tail-like form behind fast-moving dislocations,
further promoting slip localization.
Oana Cojocaru-Mirédin, Yanpeng Fu, Aleksander Kostka, Rodrigo Sáez-Araoz,
Andreas Beyer, Nikolai Knaub, Kerstin Volz, Christian-Herbert Fischer and Dierk Raabe
Prog. Photovolt: Res. Appl. 2015; 23:705–716
Prog. Photovolt Res. Appl. 2015 vol 23 p[...]
.pdf Datei [958.5 KB]
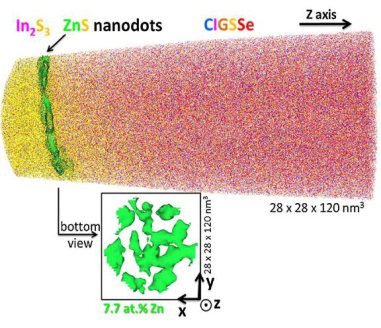
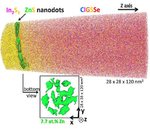
In this work, we investigate the p–n junction region for two different buffer / Cu(In,Ga)(Se,S)2 (CIGSSe) samples having different conversion efficiencies (the cell with pure In2S3 buffer
shows a lower efficiency than the nano-ZnS/In2S3 buffered one). To explain the better efficiency of the sample with nano-ZnS/In2S3 buffer layer, combined transmission electron microscopy,
atom probe tomography, and X-ray photoelectron spectroscopy studies were performed. In the pure In2S3 buffered sample, a CuIn3Se5 ordered-defect compound is observed at the CIGSSe surface,
whereas in the nano-ZnS/
In2S3 buffered sample no such compound is detected. The absence of an ordered-defect compound in the latter sample is explained either by the presence of the ZnS nanodots, which may act as a
barrier layer against Cu diffusion in CIGSSe hindering the formation of CuIn3Se5, or by the presence of Zn at the CIGSSe surface, which may disturb the formation of this ordered-defect
compound. In the nano-ZnS/In2S3 sample, Zn was found in the first monolayers of the absorber layer, which may lead to a downward band bending at the surface. This configuration is very stable
(Fermi level pinning at the
conduction band, as observed for Cd in Cu(In,Ga)Se2) and reduces the recombination rate at the interface. This effect may explain why the sample with ZnS nanodots possesses a higher efficiency.
This work demonstrates the capability of correlative transmission electron microscopy, atom probe tomography, and X-ray photoelectron spectroscopy studies in investigating buried
interfaces. The study provides essential information for understanding and modeling the p–n junction at the nanoscale in CIGSSe solar cells.
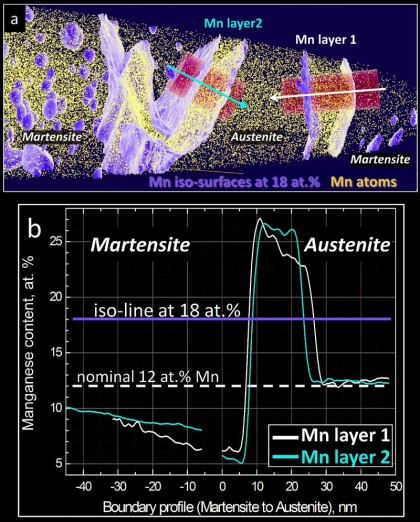
We study grain boundary embrittlement in a quenched and tempered Fe–Mn high-purity model martensite alloy using Charpy impact tests and grain boundary characterization by atom probe tomography.
Acta Materialia 86 (2015) 182 Kuzmina Gr[...]
PDF-Dokument [1.8 MB]
Here we study grain boundary embrittlement in a quenched and tempered Fe–Mn high-purity model martensite alloy using Charpy impact tests and grain boundary characterization by atom probe tomography. We observe that solute Mn directly embrittles martensite grain boundaries while reversion of martensite to austenite at high-angle grain boundaries cleans the interfaces from solute Mn by partitioning the Mn into the newly formed austenite, hence restoring impact toughness. Microalloying with B improves the impact toughness in the quenched state and delays temper embrittlement at 450 °C. Tempering at 600 °C for 1 min significantly improves the impact toughness and further tempering at lower temperature does not cause the embrittlement to return. At higher temperatures, regular austenite nucleation and growth takes place, whereas at lower temperature, Mn directly promotes its growth.
Ultramicroscopy 153 (2015) 32-39
Herbig Ultramicroscopy 153 (2015) 32 Ult[...]
PDF-Dokument [673.1 KB]
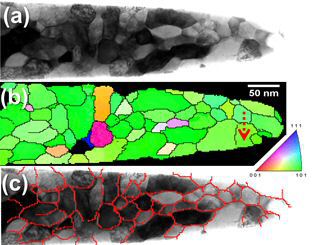
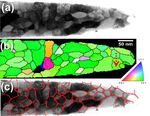
In many cases, the three-dimensional reconstructions from atom probe tomography (APT) are not sufficiently accurate to resolve crystallographic features such as lattice planes, shear bands,
stacking faults, dislocations or grain boundaries. Hence, correlative crystallographic characterization is required in addition to APT at the exact same location of the specimen. Also, for
the site-specific preparation of APT tips containing regions of interest (e.g. grain boundaries) correlative electron microscopy is often inevitable. Here we present a versatile
experimental setup that enables performing correlative focused ion beam milling, transmission electron microscopy (TEM), and APT under optimized characterization conditions. The setup was
designed for high throughput, robustness and practicability. We demonstrate that atom probe tips can be characterized by TEM in the same way as a standard TEM sample. In particular, the
use of scanning nanobeam diffraction provides valuable complementary crystallographic information when being performed on atom probe tips. This technique enables the measurement of
orientation and phase
maps as known from electron backscattering diffraction with a spatial resolution down to one nanometer.
Acta Materialia 85 (2015) 32-41
Acta Materialia 85 (2015) 32 CrN AlN lay[...]
PDF-Dokument [2.0 MB]
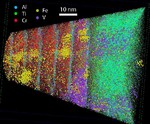
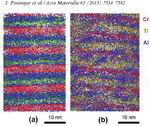
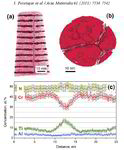
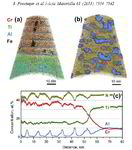
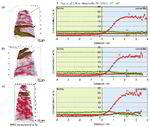
AlN/CrN superlattices with a B1 cubic crystal structure and a bilayer period of 4 nm were deposited by reactive radiofrequency magnetron sputtering. The coatings were investigated with respect to their thermal stability and changes in microstructure and chemical composition at 900 °C. The AlN layers show high chemical stability but undergo dissolution by pinching off at grain boundaries. A transformation from cubic to hexagonal AlN with subsequent coarsening at grain boundary triple junctions is observed. In contrast to AlN, the CrN layers show poor chemical stability and their compositions are shifted towards Cr2N upon annealing in a protective argon atmosphere due to nitrogen loss. However, even after establishing Cr2N stoichiometry the crystal structure of the layers remains cubic.
Acta Materialia
Volume 90, 15 May 2015, Pages 94–104
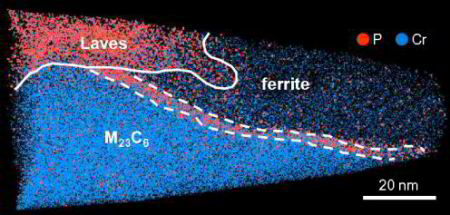
The nucleation of Mo-rich Laves phase particles adjacent to M23C6 micrograin boundary carbides in 12% Cr tempered martensite ferritic steels
M.I. Isika, A. Kostka, V.A. Yardley, K.G. Pradeep, M.J. Duarte, P.P. Choi, D. Raabe, G. Eggeler
Acta Materialia 90 (2015) 94 Laves phase[...]
PDF-Dokument [2.5 MB]
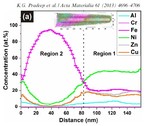
We study the nucleation of Mo-rich Laves phase particles during aging and creep of 12 wt.% Cr tempered martensite ferritic steels (TMFS). Recently, in Isik et al. (2014) we reported that Laves phase particles tend to form at micrograin boundaries of TMFSs after Mo and Si had segregated from the ferritic matrix to these internal interfaces. In the present work, we employ transmission electron microscopy (TEM) and atom probe tomography (APT) to study the formation of Laves phase particles. We investigate the preference of Laves phase particles to nucleate next to M23C6 micrograin boundary carbides. Our results suggest that this joint precipitation effect is due to the combined segregation of Mo and Si from the matrix to the micrograin boundaries and Si and P enrichment around the growing carbides.
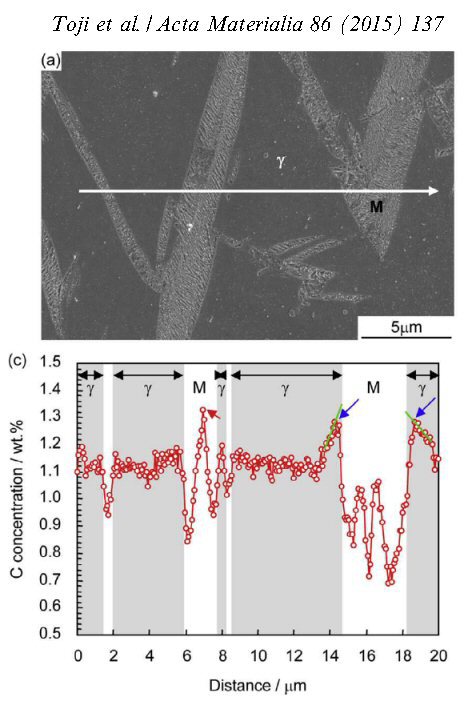
Acta Materialia 86 (2015) 137-147
Carbon partitioning during quenching and partitioning heat treatment accompanied by carbide precipitation
Yuki Toji, Goro Miyamoto, Dierk Raabe
Acta Materialia 86 (2015) 137 Toji carbo[...]
PDF-Dokument [1.8 MB]
Carbon partitioning from martensite into austenite in the quenching and partitioning (Q&P) process has been suggested to be controlled by the constrained carbon equilibrium (CCE)
criterion. It defines an approach for predicting the carbon concentration in austenite under the condition that competing reactions such as carbide formation and bainite transformation are
suppressed. Carbide precipitation in martensite is, however, often observed during the partitioning step, even in low-carbon steels as well as in high-carbon steels, even when containing a high
amount of Si. Therefore, carbon partitioning from martensite into austenite is studied here, considering carbide precipitation in martensite. Carbon partitioning was investigated by means
of a field-emission electron probe micro analysis (FE-EPMA) and atom probe tomography (APT), using 1.07 wt.% and 0.59 wt.% carbon steels with various martensite volume fractions. Carbon
partitioning from martensite to austenite was clearly observed in all specimens, even though a considerable amount of carbide precipitated inside the martensite. The austenite carbon
concentration after the partitioning step was not influenced by either the martensite volume fraction or the bulk carbon content. A modified model for predicting the austenite carbon
concentration after the partitioning step was proposed to explain the experimental results by assuming carbon equilibria between austenite,
ferrite and cementite under a constrained condition.
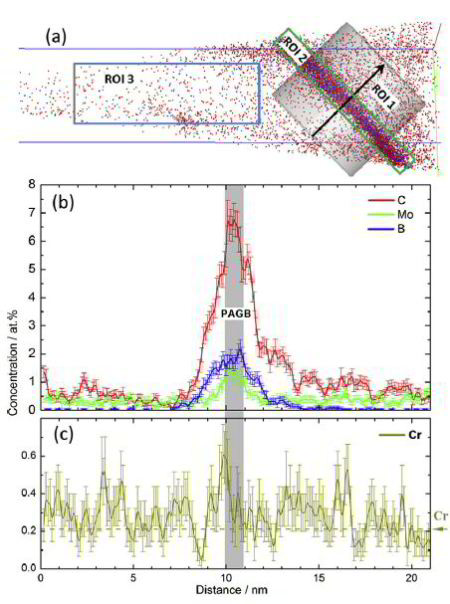
The distribution of B and other alloying elements (C, Cr, Mo) at prior austenite grain boundaries (PAGBs) and in the matrix was quantified by atom probe tomography in a quenched martensitic steel. B and Mo were observed to be segregated only at PAGBs and to be absent at martensite-martensite boundaries. C is segregated both at PAGBs and at martensite–martensite boundaries, whereas Cr is homogeneously distributed in the probed volume. Our results indicate that B undergoes a non-equilibrium segregation.
Scripta Materialia 96 (2015) 13 Li et al[...]
PDF-Dokument [1.8 MB]
Addition of a small amount of B (typically 3–30 wt ppm) to low-alloy steels can significantly enhance their hardenability [1]. The reason for such an improvement
has been frequently attributed to segregation of B at (prior) austenite grain boundaries (PAGBs), which may reduce the grain boundary energy, thus preventing the austenite to ferrite
transformation and rendering bainite or martensite formation more favorable [2,3]. Depending on heat treatment
conditions, segregation of B at grain boundaries (GBs) can be of equilibrium and/or non-equilibrium type. Equilibrium segregation [4] is driven by the difference in free enthalpy of solute
atoms in the matrix and at interfaces
such as grain boundaries. Solute segregation at grain boundaries hence thermodynamically minimizes the overall free energy of the system. Its magnitude decreases with increasing
temperature. Non-equilibrium segregation [5], on the other hand, mainly occurs upon cooling from high temperatures, during which supersaturated vacancies are induced in the matrix.
These vacancies diffuse towards vacancy sinks such as grain boundaries. They may drag solute atoms with them, leading to the non-equilibrium segregation of solutes at GBs. Therefore, in such
cases the magnitude of segregation depends on the austenitization temperature and the cooling rate [6–9]. It is widely accepted that B segregation at PAGBs during cooling is
controlled by a non-equilibrium mechanism [2,6–18]. Segregation of B at PAGBs in martensitic steels has been studied by particle tracking autoradiography (PTA) [2,9,13,16,18],
secondary ion mass spectrometry (SIMS) [9,18,19] and electron energy loss spectroscopy (EELS) [17]. These characterization techniques, except EELS, are advantageous for large-scale (up
to several hundred micrometers) investigations, providing overviews of distri-
butions of B at GBs and B-containing precipitates. However, they are not the most appropriate approaches for quantitative analyses. As a complementary technique atom probe tomography
(APT) traces individual atoms at the atomic scale, and thus delivers precise information on solute distribution at GBs, in precipitates and in the matrix. An APT study of B
distribution in a tempered martensitic steel is reported in Ref. [19]. While segregation of B at PAGBs in tempered steels was observed [19], it is still not clear whether B segregation
at PAGBs occurs during cooling (non-equilibrium segregation) or during subsequent tempering (equilibrium segregation). Also it is unclear whether B segregates to martensite–martensite
(M–M) boundaries and how other alloying elements may influence B segregation. To answer these questions, one needs to
investigate as-quenched steel. Since PAGBs are hard to reveal by chemical etching and hard to distinguish from other types of GBs such as block, packet boundaries or lath boundaries in
martensite when preparing APT samples by
focused-ion-beam (FIB) milling, investigations of B segregation at PAGBs become more difficult in a quenched steel. In the present work we apply APT to study segregation of B, C, Mo and Cr
at PAGBs and other types of boundary
in an as-quenched martensitic steel.
Guo Phys Rev Lett 113.035501 CuZr - Cu l[...]
PDF-Dokument [2.1 MB]
Deformation of ductile crystalline-amorphous nanolaminates is not well understood due to the complex interplay of interface mechanics, shear banding, and deformation-driven chemical mixing.
Here we present
indentation experiments on 10 nm nanocrystalline Cu–100 nm amorphous CuZr model multilayers to study these mechanisms down to the atomic scale. By using correlative atom probe tomography and
transmission
electron microscopy we find that crystallographic slip bands in the Cu layers coincide with noncrystallographic shear bands in the amorphous CuZr layers. Dislocations from the crystalline layers
drag Cu atoms across the interface into the CuZr layers. Also, crystalline Cu blocks are sheared into the CuZr layers. In
these sheared and thus Cu enriched zones the initially amorphous CuZr layer is rendered into an amorphous plus crystalline nanocomposite.
Andreas Stoffers, Oana Cojocaru-Mirédin, Winfried Seifert, Stefan Zaefferer, Stephan Riepe, Dierk Raabe
Grain boundary segregation in multicrystalline silicon: correlative characterization by EBSD, EBIC, and atom probe tomography
Photovoltaics: Research and Applications
http://dx.doi.org/10.1002/pip.2614
DO - 10.1002/pip.2614
2015, 1099-159X
Stoffers 2015 Photovoltaics Research and[...]
.pdf Datei [720.7 KB]
The efficiency of multicrystalline silicon solar cells suffers from the presence of extended defects like dislocations and grain boundaries. In fact, the defects themselves do
not implicitly have to be harmful, but their interaction with
impurities makes them detrimental for the cell efficiencies. Here, we present a systematic method to correlate the grain boundary charge recombination activity with local
grain boundary properties and the site specific segregation information. For that, electron beam induced current is used to characterize the recombination activity at the grain
boundaries, while electron backscatter diffraction is used to map the grain boundary crystallography. Atom probe tips containing the desired grain boundary are cut by using a novel
site-specific
sample preparation. Finally, atom probe tomography is used to reveal the 3D distribution of the impurities at the selected grain boundary. In conclusion, this work is one of the first
studies based on understanding the correlation between the charge recombination activity and structural as well as chemical properties at grain boundaries in multicrystalline silicon
solar cells.
J. Mater. Res.,
Vol. 29, No. 17, Sep 14, 2014
Materials Research Society 2014
Jaegle et al J. Mater. Res., Vol. 29 (20[...]
PDF-Dokument [489.4 KB]
Materials produced by selective laser melting (SLM) experience a thermal history that is markedly different from that encountered by conventionally produced materials. In particular, a very
high cooling rate from the melt is combined with cyclical reheating upon deposition of subsequent layers.
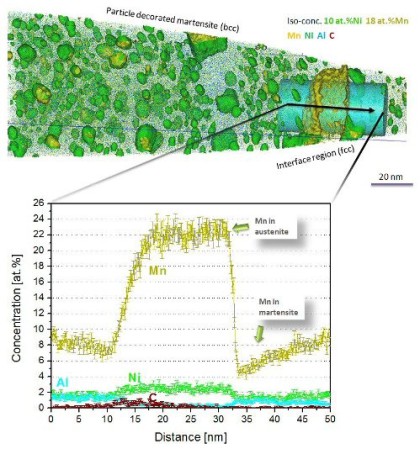
Using atom-probe tomography (APT), we investigated how this nonconventional thermal history influences the phase-transformation behavior of maraging steels
(Fe–18Ni–9Co–3.4Mo–1.2Ti) produced by SLM. We found that despite the “intrinsic heat treatment” and the known propensity of
maraging steels for rapid clustering and precipitation, the material does not show any sign of phase transformation in the as-produced state. Upon aging, three different types of precipitates,
namely (Fe,Ni,Co)3(Ti,Mo), (Fe,Ni,Co)3(Mo,Ti), and (Fe,Ni,Co)7Mo6 (l phase), were observed as well as
martensite-to-austenite reversion around regions of the retained austenite. The concentration of the newly formed phases as quantified by APT closely matches thermodynamic equilibrium
calculations.
Current Opinion in Solid State and Materials Science 18 (2014) 253-261
D. Raabe, M. Herbig, S. Sandlöbes, Y. Li, D. Tytko, M. Kuzmina, D. Ponge, P.-P. Choi
Grain boundary segregation engineering in metallic alloys: A pathway to the design of interfaces
Current Opinion in Solid State Materials[...]
PDF-Dokument [1.7 MB]
Grain boundaries (GBs) are ubiquitous defects in metallic alloys governing a range of properties, such as tensile strength, fatigue resistance, fracture toughness, strain hardening, brittleness,
conductivity, or corrosion. Unlike bulk phases they are planar and require 5 parameters to be crystallographically characterized (disorientation, plane inclination). Hence, they cannot be regarded as
scalar objects with homogeneous properties but as a high dimensional class of defects. For example, even for highly coherent GBs such as first order twins (R3 CSL (coincidence site lattice))
exponential property changes can occur if the GB plane is misaligned by only a few degrees from its most coherent position. GBs can either weaken (inter-crystalline fracture, stress corrosion
cracking) or strengthen (Hall-Petch effect) polycrystalline metallic materials. In either case the types of GBs involved affect
the specific material response substantially as was discussed particularly in the context of highly coherent versus random GBs. These observations led to the concept of grain boundary engineering
which aims at designing polycrystals with specific GB distributions. The next logical step in this context lies in also considering and engineering the segregation and its effects on GBs. This is an
obvious thought since many if not all GB properties are accessible to decoration-driven manipulation such as cohesion; energy; fracture resistance; electrical conductivity; transport coefficients;
electrochemical properties; hydrogen embrittlement; mobility; and resistance to or sources of dislocations to name only
a few effects that are relevant for structural alloys. We refer to such manipulation of GBs via solute decoration and even confined transformation as ‘grain boundary segregation engineering’ (GBSE)
or just ‘segregation engineering’. Applying GBSE needs to involve both, thermodynamic (segregation
coefficient; co-segregation; competing bulk phases) and kinetic (diffusion) influence factors. Hence, not only the associated crystallographic degrees of freedom and all segregated elements should
be
considered but also time and temperature. However, as will be addressed below, GBSE can for a number of alloys even be applied when knowing the average segregation level to non-ordered or
‘random’ GBs, i.e., a full 5D crystallographic description is not in all cases required. GB segregation changes with time when the heat treatment or application temperature is high enough for
diffusion.
Hence, also the GB properties may alter. GBSE considers solute decoration not as an undesired and inherited phenomenon but instead utilizes segregation as a tool for site-specific manipulation
that allows for optimizing specific grain boundary structures, compositions and properties that enable the design of beneficial material behavior. More specific, GB segregation can be used as a
microstructure design method since solutes affect the structure, phase state and atomic bonds within the decorated interface. Different scenarios are conceivable: first, segregation of certain
solutes might enhance GB cohesion (GB strengthening). Second, the reverse might apply, namely, reduction in cohesion and bonding strength (interface weakening) at GBs due to segregation. Third,
segregation could be strong enough, in conjunction with the available interfacial energy and local stresses, to result in a phase transformation of or at the GB region. Related to the latter point is
the possibility for the formation of complexions, i.e., interface stabilized phases. An important question in that context is the accessibility of these segregation-dependent GB features via an
‘engineering’ approach. Indeed various examples are conceivable where such misorientation- and plane-sensitive design approach can work, namely, diffusion annealing, grain growth heat treatment, and
local phase transformation effects. A typical example for an engineering heat treatment approach to tune a desired segregation level at GBs lies in a combined diffusion plus grain growth heat
treatment: Since the reduction and GB
energy may depend on both, the GB plane inclination and its crystallographic misorientation, such low energy configurations can be obtained for instance by a grain growth heat treatment, where
low-energy interfaces prevail owing to their comparably small capillary driving force. Another broadly applicable example of GB segregation engineering lies in the reduction of the average
grain
size due to an overall drop in GB energy caused by equilibrium segregation.
Although GBSE can be applied to such diverse systems as solar cells, superconductors, or ceramics we focus here on recent developments pertaining to metallic alloys used for structural
applications.
In these materials we see particularly high relevance and opportunities of the GBSE concept for designing improved ultra fine grained and nanostructured metallic alloys where GBs constitute
a large fraction of the material. Particularly alloys with nanoscaled grain size suffer often from very low ductility in conjunction with inter-crystalline damage. In such materials particularly the
internal interfaces are prone to void formation and damage initiation.
In summary grain boundaries influence mechanical, functional, and kinetic properties of metallic alloys. They can be manipulated via solute decoration enabling changes in energy, mobility, structure,
and cohesion or even promoting local phase transformation. In the approach which we refer here to as ‘segregation engineering’ solute decoration is not regarded as an undesired phenomenon but is
instead utilized to manipulate specific grain boundary structures, compositions and properties that enable useful material behavior. The underlying thermodynamics follow the adsorption isotherm.
Hence, matrix-solute combinations suited for designing interfaces in metallic alloys can be identified by considering four main aspects, namely, the segregation coefficient of the decorating element;
its effects on interface cohesion, energy, structure and mobility; its diffusion coefficient; and the free energies of competing bulk phases, precipitate phases or complexions. From a practical
perspective, segregation engineering in alloys can be usually realized by a modest diffusion heat treatment, hence, making it available in large scale manufacturing.
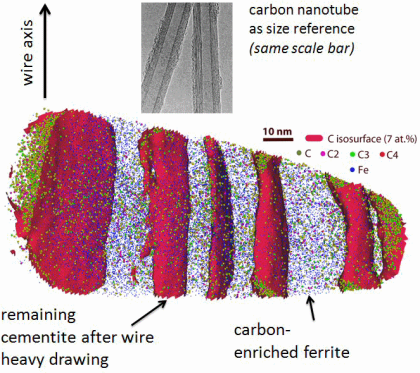
Grain refinement through severe plastic deformation enables synthesis of ultrahigh-strength nanostructured materials. Two challenges exist in that context: First, deformation-driven grain refinement is limited by dynamic dislocation recovery and crystal coarsening due to capillary driving forces; second, grain boundary sliding and hence softening occur when the grain size approaches several nanometers. Here,
both challenges have been overcome by severe drawing of a pearlitic steel wire (pearlite: lamellar structure of alternating iron and iron carbide layers). First, at large strains the carbide phase dissolves via mechanical alloying, rendering the initially two-phase pearlite structure into a carbon-supersaturated iron phase. This carbon-rich iron phase evolves into a columnar nanoscaled
Li and Raabe Phys Rev Lett vol 113 page [...]
PDF-Dokument [1.6 MB]
Acta Materialia 71 (2014) 20 metallic gl[...]
PDF-Dokument [1.5 MB]
Understanding phase changes, including their formation and evolution, is critical for the performance of functional as well as structural materials. We analyze in detail microstructural and
chemical transformations of the amorphous steel Fe50Cr15Mo14C15B6 during isothermal treatments at temperatures ranging from 550 to 800 C. By combining high-resolution transmission electron
microscopy and Rietveld analyses of X-ray diffraction patterns together with the local chemical data obtained by atom probe tomography, this research provides relevant information at the
atomic scale about the mechanisms of crystallization and the subsequent phases evolution. During the initial stages of crystallization a stable (Fe,Cr)23(C,B)6 precipitates as well as two
metastable intermediates of M3(C,B) and the intermetallic
v-phase. When full crystallization is reached, only a percolated nano-scale Cr-rich (Fe,Cr)23(C,B)6 and Mo-rich g-Fe3Mo3C structure is detected, with no evidence to suggest that other phases
appear at any subsequent time. Finally, the corrosion behavior of the developed phases is discussed from considerations of the obtained atomic information.
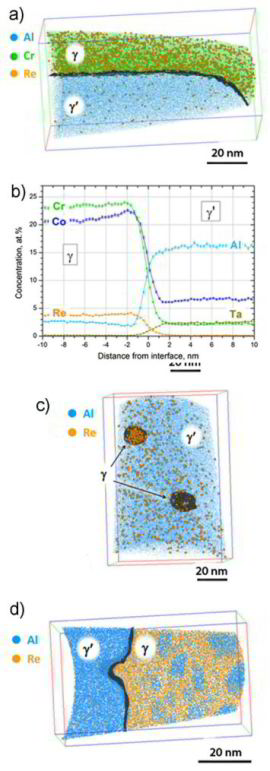
We show how conventional and advanced mechanical, chemical, and
microstructural methods can be used to characterize cast single crystal Ni-base superalloy (SX) plates across multiple length scales. Two types of microstructural heterogeneities are important, associated with the castmicrostructure (dendrites (D) and interdendritic (ID) regions – large scale heterogeneity) and with the well-known gamma/gamma' microstructure (small scale heterogeneity).Using electron probe microanalysis (EPMA),we can show that elements such as Re,Co, and Cr partition to the dendrites while ID regions contain more Al, Ta, and Ti.
Advanced Engin Mater 2014 Superalloy Mul[...]
PDF-Dokument [2.7 MB]
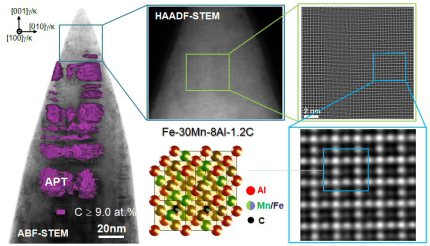
We investigate an electrolytic route for hydrogen charging of metals and its detection in Atom Probe Tomography (APT) experiments. We charge an austenitic Fe-30Mn-8Al-1.2C (wt.%) weight reduced high-Mn steel and subsequently demonstrate the detectability of deuterium in an APT experiment. The experiment is repeated with a deposited Ag film upon an APT tip of a high-Mn steel. It is shown that a detectable deuterium signal can be seen in the high-Mn steel, and a D:H ratio of 0.84 can be reached in Ag films. Additionally, it was found that the predicted time constraint on detectability of D in APT was found to be lower than predicted by bulk diffusion for the high-Mn steel.
Intern Journal Hydrogen Energy v...2014)[...]
PDF-Dokument [541.6 KB]
B2 NiMn and Ni2MnAl Heusler nanoprecipitates are designed via elastic misfit stabilization in Fe–Mn maraging steels by combining transmission electron microscopy (TEM) correlated atom probe tomography (APT) with ab initio simulations. Guided by these predictions, the Al content of the alloys is systematically varied, and the influence of the Al concentration on structure stability, size and distribution of precipitates formed during ageing at 450 C is studied using scanning electron microscopy–electron backscatter diffraction, TEM and APT.
Acta Mater 2014 Fe–Mn maraging steels wi[...]
PDF-Dokument [2.6 MB]
Phys Rev Lett. 2014 grain boundary segre[...]
PDF-Dokument [842.8 KB]
Grain boundaries influence mechanical, functional, and kinetic properties of metallic alloys. They can be manipulated via solute decoration enabling changes in energy, mobility, structure, and cohesion or even promoting local phase transformation. In the approach which we refer here to as ‘segregation engineering’ solute decoration is not regarded as an undesired phenomenon but is instead utilized to manipulate specific grain boundary structures, compositions and properties that enable useful material behavior. The underlying thermodynamics follow the adsorption isotherm. Hence, matrix-solute combinations suited for designing interfaces in metallic alloys can be identified by considering four main aspects, namely, the segregation coefficient of the decorating element; its effects on in
Current Opinion 2014 Grain Boundary Segr[...]
PDF-Dokument [1.7 MB]
METALLURGICAL AND MATERIALS TRANSACTIONS A VOLUME 45A, FEBRUARY (2014) 595
WENWEN SONG, PYUCK-PA CHOI, GERHARD INDEN, ULRICH PRAHL, DIERK RAABE, and WOLFGANG BLECK
Partitioning carbide atom probe tomograp[...]
PDF-Dokument [1.1 MB]
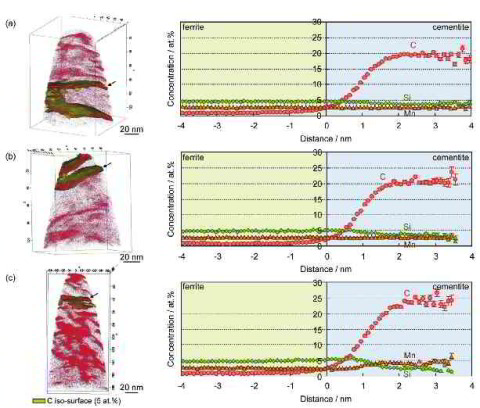
In this paper we report about the characterization of high carbon bearing steel 100Cr6 using electron microscopy and atom probe tomography in combination with multi-component diffusion
simulations. Scanning electron micrographs show that around 14 vol pct spheroidized carbides are formed during soft annealing and only 3 vol pct remain after dissolution into the austenitic
matrix through austenitization at 1123 K (850 C) for 300 seconds. The spheroidized particles are identified as (Fe, Cr)3C by transmission electron microscopy. Atom probe analysis reveals
the redistribution and partitioning of the elements involved, i.e., C, Si, Mn, Cr, Fe, in both, the spheroidized carbides and the bainitic matrix in the sample isothermally heat-treated at
773 K (500 C) after austenitization. Homogeneous distribution of C and a Cr gradient were detected within the spheroidized carbides. Due to its limited diffusivity in (Fe, Cr)3C, Cr
exhibits a
maximum concentration at the surface of spheroidized carbides (16 at. pct) and decreases gradually from the surface towards the core down to about 2 at. pct. The atom probe results also
indicate that the partially dissolved spheroidized carbides during austenitization may serve as nucleation sites for intermediate temperature cementite within bainite, which results in
a relatively softer surface and harder core in spheroidized particles. This microstructure may contribute to the good wear resistance and fatigue properties of the steel. Good
agreement
between DICTRA simulations and experimental composition profiles is obtained by an increase of mobility of the substitutional elements in cementite by a factor of five, compared to
the mobility in the database MOBFE2.
2012 atom probe analysis of compound sem[...]
PDF-Dokument [1.9 MB]
Carbon partitioning between ferritic and austenitic phases is essential for austenite stabilization in the most advanced steels such as those produced by the quenching and partitioning (Q&P) process.
Acta Mater 2014 carbon partitioning mart[...]
PDF-Dokument [4.0 MB]
We report on the characterization of grain boundary (GB) segregation in an Fe–28Mn–0.3C (wt.%) twinning-induced plasticity (TWIP) steel. After recrystallization of this steel for 24 h at 700 C, 50% general grain boundaries (GBs) and 35% R3 annealing twin boundaries were observed (others were high-order R and low-angle GBs). The segregation of B, C and P and traces of Si and Cu were detected at the general GB by atom probe tomography (APT) and quantified using ladder diagrams. In the case of the R3 coherent annealing twin, it was necessary to first locate the position of the boundary by density analysis of the atom probe data, then small amounts of B, Si and P segregation and, surprisingly, depletion of C were detected. The concentration of Mn was constant across the interface for both
Acta Mater 2014 GB segregation twin TWIP[...]
PDF-Dokument [665.4 KB]
Rapid annealing induced crystallization of soft magnetic Fe–Si nanocrystals in a Fe73.5Si15.5Cu1Nb3B7 amorphous alloy has been systematically studied by atom probe tomography in comparison with conv
Acta Mater FeSiNbBCu nanocrystalline mag[...]
PDF-Dokument [1.6 MB]
Carbon-supersaturated nanocrystalline hypereutectoid steels with a tensile strength of 6.35 GPa were produced from severely cold-drawn pearlite. The nanocrystalline material undergoes softening upon annealing at temperatures between 200 and 450°C. The ductility in terms of elongation to failure exhibits a non-monotonic dependence on temperature.
Acta Materialia 84 (2015) 110-123 atom p[...]
PDF-Dokument [2.4 MB]
Image Gallery on Atom probe Tomography
Atom Probe Tomographic Analysis of Corrosion
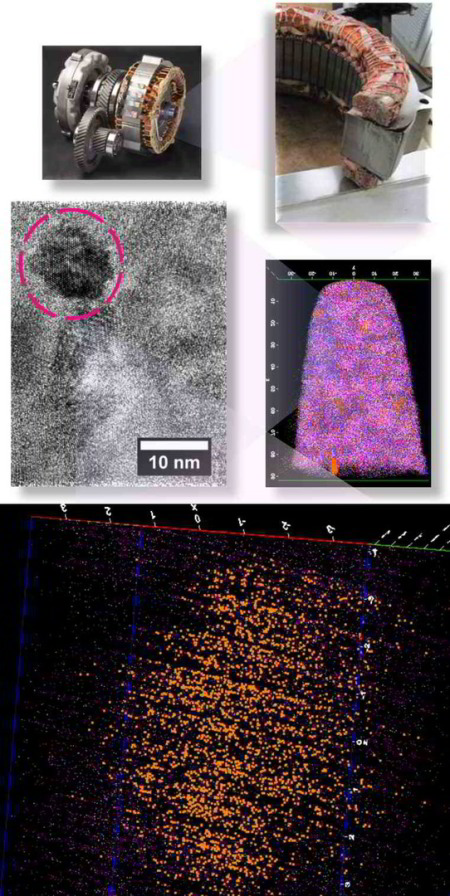
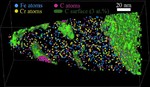
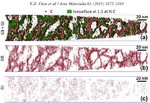
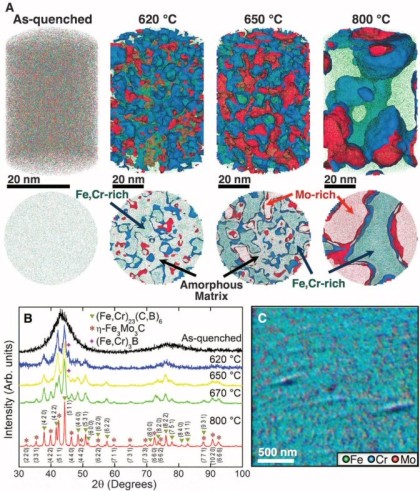
Corrosion destroys 75 billion euros of economic value each year in Germany alone. Soon it may be possible to better assess which steels and alloys will be affected less to corrosion, and how to limit the overall damage: We recently analyzed an amorphous steel comprising iron, chromium, molybdenum, boron, and carbon. We observed that the more ordered the material’s structure is, and the more uneven the distribution of its atoms, the more easily it is corroded by rust. If the elements of the alloy do not form a regular crystal lattice and are distributed completely uniformly across the material, then, under corrosive conditions, a passivation layer forms on its surface and protects it from rusting. If, in contrast, ordered nanocrystals form that sometimes contain more chromium and sometimes more molybdenum, the corrosion quickly eats away the material because no protective passivation layer forms. These findings give materials scientists clues as to what they should pay attention to regarding the composition and production of materials.
In humid air, chemistry gnaws at every metal, unless it happens to be gold or another precious metal. But oxygen in the air is only too happy to hook up with the less precious representatives of this class of materials. Humidity helps it in its search for a partner, and all the more so the saltier it is. At the outset of a corrosive attack, some metals, such as chromium and zinc, become coated with a dense oxide layer that prevents further advances on the part of the oxygen. If no such passivation layer forms, the metal corrodes, leaving behind a porous and brittle surface that causes a car body or a ship's hull to gradually crumble. This is how corrosion destroys up to 4 percent of the economic output of the industrialized nations every year.
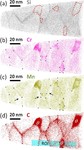
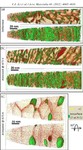
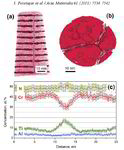
We investigated in detail how the micro- or nanostructure of a Fe50Cr15Mo14C15B6 steel influences its corrosion resistance. This alloy is one of the amorphous steels that are used, for example, as abrasion-resistant coatings. The elements in it are present in an irregular structure, but they are distributed completely uniformly in the material. Here we were specifically interested in this alloy primarily as a model system in which we can study the transition from the amorphous form to the crystalline form. Since it is similar to common chromium-molybdenum steels, our findings are also relevant for materials used in actual practice. The test alloy does, however, contain significantly more carbon than common steels, and also the high boron content is rather unusual for steels from the blast furnace. However, this was the mixture that allowed the team to produce the amorphous variant of the alloy.
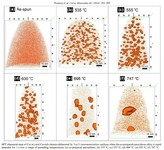
The amorphous form of the alloy marked the starting point of a series of measurements that led, via multiple intermediate stages, to the ordered crystalline form. The extent to which the alloy crystallizes depends on the temperature at which the researchers bake the elements together: the material remains amorphous at 550°C, while it takes on the completely crystalline form at 800°. Scientists obtained the intermediate stages at 620–670°C.
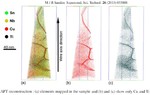
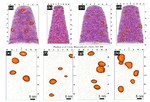
We then used atom probe tomography to reveal the nanostructures of the steel variants. In this process, the atoms of a sample were evaporated one by one and collected by a detector. The element can be deduced from the time it takes an atom to reach the detector, and its position in the probe from the point of impact. This allowed us to obtain precise maps of the materials, and the maps helped them understand how the atoms regroup with increasing manufacturing temperature. Different regions form, in each of which one of the metals is concentrated. The sample that we produced at 620°C remains largely disordered. But crystalline structures can already be seen in this sample, too. In contrast, the form produced at 800° is completely crystalline, with each of the regions extending up to 50 nm only.
We next compared the precise information about the nanostructure of the steel variants with electrochemical measurements to assess how easily the various forms fall victim to corrosion. To do this, they wetted the sample surfaces with a saline solution and applied an electrical voltage to the alloy. The higher the voltages a sample withstands in the lab without corroding, the more resistance it shows to the rust attack in practice, too. The completely amorphous and the largely amorphous forms proved to be almost as resistant as pure chromium. Under normal conditions, corrosion stands little chance against chromium, because it is sealed by a thin passivation oxide layer. Such a protective layer also forms on the amorphous steels. In the largely crystalline forms of the alloy, however, that is apparently no longer possible. Here, the rust already eats holes in the sample at relatively low voltages.
Science 341, 372 (2013)
Science-2013-Duarte-372-6.pdf
PDF-Dokument [1.7 MB]
Atom Probe Instrumentation at the Max-Planck Institute
In our group we use a state-of-the-art three-dimensional atom probe which was installed in February 2010 at the Department for Microstructure Physics and Alloy Design at the Max-Planck Institut für Eisenforschung in Düsseldorf. This unique atomic-scale resolving characterization tool enables spatially resolved chemical analyses of nanostructured materials with sub-nanometer resolution (x = y ≈ 0.2 nm and z ≈ 0.1 nm). By applying either high voltage or laser pulses to a positively biased tip-shaped needle specimen (having a radius of curvature <100 nm), surface atoms are successively ionized and field-evaporated. The field-evaporated ions are accelerated towards a position sensitive detector that records their time of flight (TOF) and impact positions. A three-dimensional elemental map is reconstructed from the collected data using a point projection algorithm. The instrument we use in our department (Imago LEAP 3000X HR) is equipped with a local electrode, a wide-angle reflectron, a high-speed delay line detector system as well as an ultrafast laser with a pulse width of 10 ps and wavelength of 510 nm. Such an instrument design has numerous advantages over conventional atom probes. The local electrode enhances the electric field at the specimen and allows fast pulsing (max. 200 kHz) with low voltage pulses. A high-speed delay line detector system provides fast data acquisition rates of up to 2 Mio ions/min. Due to the proximity between the specimen and detector, the field of view can be as large as 200 nm. As a result, large volumes can be probed within a few hours, which can contain up to several hundred millions of atoms. The wide-angle reflectron substantially enhances the mass resolution of this instrument to a value of dm/m=1100 (FWHM at m/n = 27). Complex multi-component systems can therefore be analyzed with ease and high compositional accuracy. Furthermore, impurity concentrations as low as few tens of ppm can be detected. The ultra-fast laser extends the applicability of this technique to materials having low electrical conductivity such as semiconductors and ceramics.
Owing to its outstanding performance, the spectrum of materials currently investigated by this new instrument is wide, ranging from binary model systems for fundamental studies, complex engineering alloys (ultrahigh-strength steels, Ni-based superalloys), wear-resistant ceramic hardcoatings (transition metals nitride multilayers) to semiconductor devices (thin film solar cells), etc.. Some of the on-going projects of the atom probe group at the department are summarized below:
A major research topic is the alloying process of nanocomposites (Cu-Fe, Cu-Nb, Fe/Fe3C) driven by severe plastic deformation. Using the atom probe, the level of interfacial mixing and bulk
supersaturation of ball-milled and wire-drawn samples is determined. Applying high-resolution electron microscopy as a complementary technique, detailed insights can be gained into the transformation
of a nanostructured material upon plastic deformation. Another project deals with the development of precipitation hardened ductile high strength martensitic and austenitic-martensitic steels. The
alloys are characterized by low carbon contents and various Mn concentrations to achieve different levels of austenite stability. Further elements including Ni, Al, Si, Ti and Mo are added to form
nano-precipitates during an ageing heat treatment. Atom probe yields important information on these precipitates such as chemical composition, morphology, size, and number density and helps to
understand the outstanding age-hardening behavior of this maraging steel. Besides the characterization of nanoscale precipitates, the strength of the atom probe technique lies in the compositional
analysis of internal interfaces. Interface composition and width control the mechanical and tribological properties of transition metal nitride multilayers, which are applied as wear-resistant
hardcoatings for cutting-tools. The structure-property relationship and thermal stability of a high-performance hardcoating system (AlN/CrN) is currently under investigation. The atom probe group
also deals with the functional properties of advanced thin film systems. The photovoltaic properties of CuInGaSe2 and CdTe thin film solar cells are believed to be strongly dependent on the chemical
gradients across their p/n junctions and the concentration and distribution of dopant atoms (Na, Fe in CuInGaSe2, Cl in CdTe). It is our aim to elucidate these relationships by atom probe
analyses.
The Imago LEAP 3000X HR atom probe complements the diffraction-based analytical instruments at the Department for
Microstructure Physics and Alloy Design including HRTEM (Jeol JEM-2200F) and 3D EBSD (LEO SFEG SEM).
the tomographic atom probe provides fundamental new insights for advanced alloy design by probing the chemcial nature of single atoms in complex engineering materials.
Atom probe samples can be prepared by electrochemical polishing or focused ion beam and subsequent further sharpening using a focused ion beam device. Pulsed-laser tomography is in our group at the Max-Planck Institut in Düsseldorf performed using a local electrode atom probe (LEAPTM 3000X HR, Imago Scientific Instruments) tomograph at a specimen temperature in the range between 20 K and 60 K. An ultrafast pulsed laser of ~ 10 ps pulse width and 532 nm wavelength can be applied at a frequency of 250 kHz. The detection rate (target evaporation rate) usually amounts for steels to 5 atoms per 1000 pulses. Data analysis is performed using the IVAS® software from Imago Scientific Instruments. Details are given at our MPI website.
Mechanical alloying and amorphization in Cu–Nb–Ag in situ composite wires studied by transmission electron microscopy and atom probe tomography
Acta Mater 57(2009)5254.pdf
PDF-Dokument [1.7 MB]
pearlite_decomposition_Acta_Materialia_2[...]
PDF-Dokument [2.8 MB]
Metals_at_extremes-MRS_Bulletin-Dec2010_[...]
PDF-Dokument [5.5 MB]
PDF-Dokument [2.1 MB]
Atom probe tomography (APT) and steels
Atom probe tomography (APT) has essentially contributed to our understanding and development of structural materials through the analysis of solute behavior, cluster formation, precipitate
evolution, and interfacial and grain boundary chemistry at the atomic scale. Whether one is concerned with light alloys, Ni-based superalloys, or steels, the design objectives are similar, namely,
developing alloys with optimum properties (strength, toughness, ductility, fatigue
resistance, creep strength) through controlled solute solution, precipitation, grain structure, and design and combination of phases.
Performance in service, through microstructural stability and resistance to degradation, is also a major design criterion for the development of novel materials.
The ability to probe the atomic-scale structure of complex structural alloys has been instrumental in providing key insights into the atomistic mechanisms controlling microstructure, in providing routes for the development of better alloys and in suggesting solutions to remediate problematic degradation phenomena.
We are particularly interested on the impact that APT has had on our understanding of steels, from the development of new strengthening strategies to designing materials more resistant to degradation. The examples we work on illustrate the power of APT in atom-by-atom analysis, three-dimensional reconstruction, and equal sensitivity for all chemical species, including light elements, such as carbon and nitrogen.
Interface-directed spinodal decomposition in TiAlN/CrN multilayer hard coatings studied by atom probe tomography
APT-TiAlN-CrN multilaerys Acta-2013.pdf
PDF-Dokument [3.5 MB]
2012_Steel_Atom-probe_Microscopy-Today.p[...]
PDF-Dokument [1.4 MB]
We study the structure and chemical composition of the j-carbide formed as a result of isothermal transformation in an Fe–3.0Mn–5.5Al–0.3C alloy using transmission electron microscopy and atom p
Scripta-kappa-carbides-in-steel.pdf
PDF-Dokument [1.0 MB]
Spheroidized carbide atom probe tomograp[...]
PDF-Dokument [1.1 MB]
Atom probe tomography study of corrosion at the single element level
Science-2013-Duarte-372-6.pdf
PDF-Dokument [1.7 MB]
Atom probe tomography on a superplastic steel
Excellent superplasticity (elongation 720%) is observed in a novel multi-component (Mn–S–Cr–Al alloyed) ultrahigh carbon steel during tensile testing at a strain rate of 2 10^-3 s1 and a temper
Acta-Mat-2013-superplastic-steel-Al-cont[...]
PDF-Dokument [6.0 MB]