Green Steel by Ammonia Based Direct Reduction of Iron Oxides: Sustainable Steel
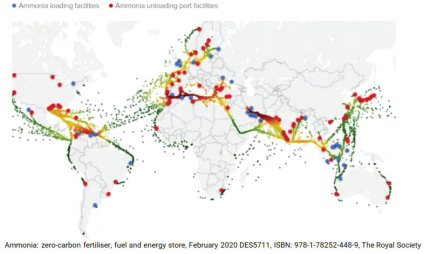
Iron making is the biggest single cause of global warming. The reduction of iron ores with carbon generates about 7% of the global carbon dioxide emissions to produce ≈1.85 billion tons of steel per year. This dramatic scenario fuels efforts to re-invent this sector by using renewable and carbon-free reductants and electricity. Here, the authors show how to make sustainable steel by reducing solid iron oxides with hydrogen released from ammonia. Ammonia is an annually 180 million ton traded chemical energy carrier, with established transcontinental logistics and low liquefaction costs. It can be synthesized with green hydrogen and release hydrogen again through the reduction reaction. This advantage connects it with green iron making, for replacing fossil reductants. the authors show that ammonia-based reduction of iron oxide proceeds through an autocatalytic reaction, is kinetically as effective as hydrogen-based direct reduction, yields the same metallization, and can be industrially realized with existing technologies. The produced iron/iron nitride mixture can be subsequently melted in an electric arc furnace (or co-charged into a converter) to adjust the chemical composition to the target steel grades. A novel approach is thus presented to deploying intermittent renewable energy, mediated by green ammonia, for a disruptive technology transition toward sustainable iron making.
https://doi.org/10.1002/advs.202300111
Advanced Science - 2023 - Ma - Reducing [...]
PDF-Dokument [2.1 MB]
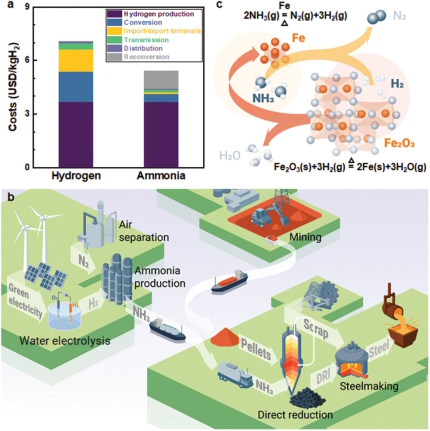
The method consists in producing green steel by using ammonia (produced from green hydrogen) as a reducing agent.
Here are the main steps to make green steel with ammonia:
The first step is to produce the usual iron ore concentrate (e g pellets or fine ores etc.).
The iron ore concentrate is then heated in a furnace.
To produce green steel, ammonia is next used as a reducing agent instead of coal (producing the reducing CO).
The ammonia is mixed with the iron ore concentrate and heated in a furnace. The ammonia reacts with the oxygen in the iron oxide, producing nitrogen gas (N2) and water (H2O). This reaction releases hydrogen gas (H2), which reacts with the remaining iron oxide to produce iron.
The iron produced from the ammonia reduction process is then refined into steel using various techniques, such as the usualy electric arc furnace and ladle secondary metallurgy methods.
What is ammonia and why can it serve in green steel making?
Ammonia is a colorless, pungent gas that consists of one nitrogen atom and three hydrogen atoms, with the chemical formula NH3. It is a common compound used in various industrial processes, including fertilizer production, refrigeration, and as a cleaning agent. Ammonia is also used as a reducing agent in some green steel production processes, as it can help to reduce greenhouse gas emissions.
Ammonia has a strong odor and is highly soluble in water, which makes it a useful compound for many applications. It is commonly used in household cleaning products and is also used in the manufacture of plastics, fibers, and explosives. In addition to its industrial uses, ammonia is also produced naturally by some organisms, such as bacteria and fish, and is found in trace amounts in the atmosphere.
https://onlinelibrary.wiley.com/doi/10.1002/advs.202300111
Iron making is the biggest single cause of global warming. The reduction of iron ores with carbon generates about 7% of the global carbon dioxide emissions to produce ≈1.85 billion tons of steel per year. This dramatic scenario fuels efforts to re-invent this sector by using renewable and carbon-free reductants and electricity. Here, the authors show how to make sustainable steel by reducing solid iron oxides with hydrogen released from ammonia. Ammonia is an annually 180 million ton traded chemical energy carrier, with established transcontinental logistics and low liquefaction costs. It can be synthesized with green hydrogen and release hydrogen again through the reduction reaction. This advantage connects it with green iron m
Green steel via hydrogen based direct re[...]
PDF-Dokument [3.3 MB]
Why do we need green steel?
We need green steel to reduce the carbon footprint of the steel industry, which is one of the largest contributors to greenhouse gas emissions worldwide. According to the International Energy Agency (IEA), the iron and steel industry is responsible for about 8% of global carbon dioxide emissions.
Green steel is produced using low-carbon technologies that reduce or eliminate greenhouse gas emissions during the steelmaking process. By using renewable energy sources, such as wind and solar power, and by using hydrogen or other low-carbon reducing agents instead of coal, green steel can significantly reduce the carbon footprint of the steel industry.
In addition to reducing greenhouse gas emissions, green steel also offers economic benefits. As governments around the world adopt more stringent climate policies and impose carbon taxes, companies that produce green steel may have a competitive advantage over those that continue to rely on high-emission production methods. Furthermore, green steel can help to meet the growing demand for sustainable products and materials among environmentally conscious consumers.
Overall, green steel is an important step towards achieving a more sustainable and environmentally friendly future, and it has the potential to significantly reduce the carbon footprint of the steel industry.
Green steel from ammonia-based direct reduction: Take home
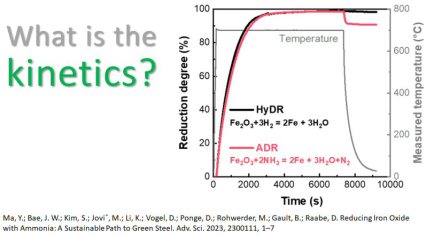
In essence we have proven that ammonia-based direct reduction (ADR; via the hydrogen bound in it and releaed from it) is kinetically as effective for producing green iron as HyDR at 700 °C. The direct utilization of ammonia in the reduction process offers a process shortcut, alleviating the need for a preliminary ammonia cracking step into hydrogen and nitrogen. During the redox reaction, the gradually generated porous iron further catalyses the decomposition of ammonia at elevated temperatures, to release hydrogen for the reduction of iron oxides. This autocatalytic reaction provides a path to further efficiency gains and cost reductions. The in-situ nitriding from the process offers protection of the pure iron against environmental degradation that otherwise requires dedicated additional process steps that are energetically and logistically costly. Such a protective nitride phase can be completely dissolved and removed after a subsequent melting process. Thus, ADR provides a novel approach to deploying intermittent renewable energy for an unprecedented and disruptive technology transition toward sustainable metallurgical processes. With these benefits, it connects two of the currently most greenhouse gas intense industries (namely, steel and ammonia production industries) and opens a pathway to render them more environmentally benign and sustainable. At the same time, it can eliminate logistic and energetic disadvantages associated with the use of pure hydrogen, when it needs to be transported.