Ammonia as Reductant and Alloying Reactant in Sustainable Iron Oxide Reduction: A Unified Metallurgical Perspective
Ammonia: From Hydrogen Carrier to Process Gas
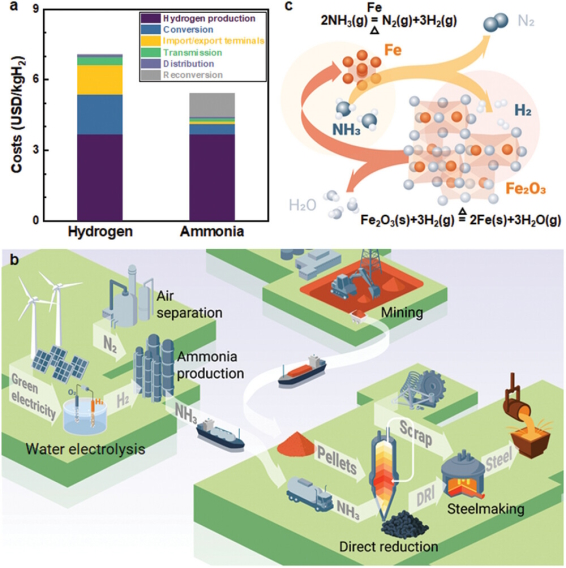
The global steel industry emits approximately 3.73 billion tonnes of CO₂ per year, with roughly 79% of those emissions originating from the blast furnace–basic oxygen furnace route that converts iron ores into pig iron through fossil carbon reductants . Decarbonizing this sector requires not merely incremental improvements to existing infrastructure but a genuine paradigm shift in how the reduction chemistry is conceived at the molecular level. Hydrogen-based direct reduction (HyDR) has emerged as one of the most mature alternatives, currently operating at technology readiness levels of 6–8 . Yet the deployment of HyDR at intercontinental scale runs into the long-standing logistical constraints of hydrogen: its storage demands pressures of 350–700 bar or temperatures below −253 °C, and the energy penalty for reaching these conditions consumes more than 30% of the embodied chemical energy it delivers . These are not minor inconveniences but fundamental engineering barriers that threaten the cost competitiveness of an otherwise sound chemical approach.
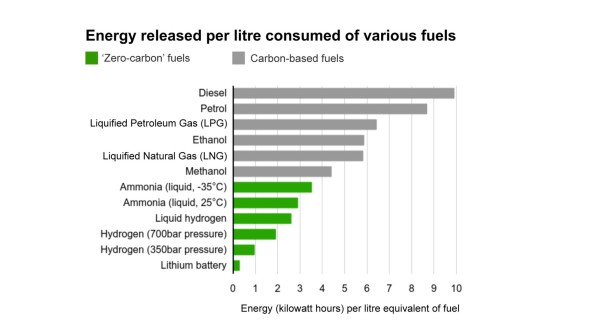
Ammonia (NH₃) offers a physically and thermodynamically more tractable alternative as an energy and hydrogen carrier. Its volumetric hydrogen content of approximately 121 kg-H₂ m⁻³ significantly surpasses that of liquid hydrogen at 70.8 kg-H₂ m⁻³, and its energy density of 4.25 kWh L⁻¹ compares favorably with liquid hydrogen at 2.81 kWh L⁻¹ . Perhaps more practically, ammonia can be liquefied at just 8 bar at room temperature or at −33 °C at atmospheric pressure, conditions for which robust and globally distributed infrastructure already exists . Projections from the International Energy Agency place the delivered cost of green ammonia in 2030 at approximately 5.5 USD kg⁻¹ H₂ (for a representative Australia-to-Japan scenario), compared with 7.1 USD kg⁻¹ H₂ for compressed or liquefied green hydrogen along the same route . Crucially, the direct utilization of ammonia in reduction furnaces—bypassing the preliminary catalytic cracking step—lowers that figure further to approximately 4.5 USD kg⁻¹ H₂ . These economic arguments alone would justify serious investigation of ammonia-based direct reduction (ADR), but the metallurgical reality turns out to be considerably richer: ammonia is not merely a hydrogen vehicle but an active reactant that simultaneously reduces, nitrifies, and, as will be discussed, alloys the iron-containing substrate.
Reduction Kinetics and the Autocatalytic Mechanism
The reduction behavior of hematite pellets under pure ammonia atmospheres at 700 °C was investigated in detail by Ma et al. using thermogravimetry coupled with quadrupole mass spectrometry. Three distinct stages were identified. Below approximately 350 °C, the gas atmosphere remains essentially unchanged. Between 350 and 650 °C, a marked decline in the NH₃ signal and a simultaneous rise in the H₂ derivative signal indicate the onset of ammonia decomposition at the oxide surface. The third and decisive stage begins above roughly 650 °C, when the freshly generated hydrogen drives the actual oxygen-removal redox reactions, with the reduction degree climbing steeply and ultimately reaching approximately 98%—a level statistically indistinguishable from HyDR under equivalent conditions . Crucially, as metallic iron nucleates and grows within the porous pellet, the catalytic efficiency of the surface for ammonia cracking increases markedly, since metallic iron is a far more active catalyst for NH₃ decomposition than iron oxides. This creates a self-reinforcing, autocatalytic loop: the iron produced by the reduction reaction further accelerates the decomposition of ammonia, providing ever more hydrogen for further reduction . The practical consequence is that no external precious-metal catalytic cracker (e.g., ruthenium-based) is required upstream of the shaft furnace, substantially reducing capital and operating costs.
The temperature dependence of ADR kinetics was mapped systematically by Jovičević-Klug et al. across the range of 500–800 °C. At 700 °C, the reduction curves for ADR and HyDR are virtually superimposable during isothermal holding. However, at lower temperatures, important divergences emerge: at 500 °C, the kinetics of ADR are substantially slower than those of HyDR, primarily because the decomposition of ammonia into reactive hydrogen species is kinetically limited below ~550 °C . The degree of decomposition is not a simple step function of temperature; both iron oxides and the nascent metallic iron act as catalysts that lower the cracking temperature to approximately 300–350 °C, yet the conversion efficiency only approaches complete decomposition above 600 °C . This means that at lower process temperatures, a fraction of the gas in contact with the pellet is still NH₃ rather than H₂, and simultaneous nitriding of the partially reduced material takes place concurrently with oxygen removal, effectively competing with—and in some regimes retarding—the reduction reaction. The gas evolution analysis performed by Jovičević-Klug et al. using a quadrupole mass spectrometer placed in close proximity to the sample during TGA confirmed that the H₂/N₂ signal ratio evolves distinctly at different temperatures, reflecting these competing reaction pathways . A particularly instructive diagnostic is the water signal, which mirrors the instantaneous oxygen removal rate and was proposed as a practical tool for on-line process monitoring of the reduction degree in an industrial shaft furnace environment.
Nitriding as an Intrinsic Consequence and a Metallurgical Opportunity
Perhaps the most scientifically intriguing—and industrially consequential—aspect of ADR is the inevitability of nitrogen incorporation into the reduced product. This occurs through two mechanistically distinct pathways. In-process nitriding happens simultaneously with oxygen removal during the isothermal reduction step: at temperatures below 600 °C, in particular, nitrogen-containing species from ammonia decomposition compete with hydrogen for interaction with the iron surface, leading to the formation of iron nitrides (primarily Fe₂N and Fe₃N) within the outer shell of the pellet while the core remains insufficiently reduced . Cross-sectional XRD line-scan analysis demonstrated a characteristic shell–core architecture in pellets reduced at 500 °C: iron nitrides dominate the outer regions where metallic iron is most available for nitriding, while unreduced oxide phases persist in the interior . As the reduction temperature increases to 550–600 °C, Fe₄N becomes the dominant nitride, consistent with the thermodynamic stability of the Fe–N phase diagram at these temperatures.
The second nitriding pathway—spontaneous nitriding—occurs during cooling of the fully or partially reduced pellet under the ammonia atmosphere. This phenomenon was documented in the foundational ADR study by Ma et al. as an apparent decline in the reduction degree of approximately 7.5% during cooling from 700 °C, corresponding to a nitrogen uptake of approximately 3.3 wt% as measured by ICP-OES. The XRD and EBSD analyses of the ADR product confirmed a dual-phase microstructure of approximately 60 wt% α-iron and 40 wt% Fe₄N . Atom probe tomography (APT) measurements at near-atomic resolution revealed that the Fe₄N nitride contained approximately 21 at% nitrogen at its stoichiometric core, while a transition zone extending over 100 nm into the iron beneath the nitride interface contained approximately 6 at% nitrogen . Jovičević-Klug et al. showed that the magnitude of spontaneous nitriding is strongly coupled to the reduction degree achieved before cooling begins: a higher degree of metallization—and therefore a greater amount of catalytically active metallic iron surface—amplifies the nitriding rate during the cooling excursion. Interestingly, the effect peaks at intermediate temperatures around 600 °C and declines at 800 °C, suggesting a complex interplay of thermodynamic driving force, surface kinetics, and the evolving P(NH₃)/P(H₂) ratio as the atmosphere cools . A straightforward engineering countermeasure is available: purging the atmosphere with inert argon before cooling eliminates the nitrogen source, suppressing spontaneous nitriding and yielding a product with low residual nitrogen content . This provides a direct process lever for tuning the nitrogen content in the final product.
Far from being an unwanted side effect, the formation of Fe₄N on the surface of the reduced pellets carries significant practical advantages. The nitride layer acts as a passivating barrier against reoxidation—a critical safety hazard with conventional DRI, whose highly porous morphology (typically >40 vol% open porosity) renders the freshly reduced iron pyrophoric in humid air . The nitrogen-passivated ADR product resists reoxidation far more effectively than its HyDR counterpart, potentially obviating the need for hot briquetting into hot briquetted iron (HBI) for safe transport and storage . Upon subsequent melting in an electric arc furnace, the Fe₄N phase dissolves completely, and the nitrogen content of the final liquid drops to 0.1–0.15 wt% retained in solid solution, well within the specification range for many commercial steel grades.
From Reduction to Advanced Alloy Design: The Reactive Vapor-Phase Dealloying–Alloying Paradigm
The conceptual leap from ammonia as a reducing gas to ammonia as a simultaneous alloy design tool was made explicit in the work of Wei et al. . Their study introduced a reactive vapor-phase dealloying–alloying synthesis route that integrates, in a single solid-state operation, four principal metallurgical processes: (i) reactive dealloying through oxide reduction, (ii) substitutional alloying by interdiffusion among metallic species, (iii) interstitial alloying through nitrogen uptake from the vapor phase, and (iv) phase transformation–driven nanostructuring. The NH₃ molecule is assigned a precisely dualistic role: the hydrogen it releases upon decomposition serves as the reductant for oxygen removal, while the nitrogen it yields is consciously harvested as an interstitial alloying element.
The experimental demonstration employed cold-compacted Fe₂O₃–NiO pellets targeting an Fe–10 at% Ni stoichiometry, processed under pure NH₃ at temperatures above 455 °C . In situ synchrotron X-ray diffraction during continuous heating revealed a progression of phase events: the sequential decomposition of NiO and Fe₂O₃, the transient formation of iron nitride phases including Fe₃N and (Fe,Ni)₄N, and their eventual decomposition into a metallic FCC+BCC solid solution at higher temperatures . The resulting bulk alloy exhibited both FCC (austenite) and BCC (ferrite/martensite) phases, with Rietveld-refined lattice parameters of 3.60 and 2.87 Å respectively—both slightly expanded relative to pure Ni and pure Fe, providing direct crystallographic evidence of substitutional alloying . The microstructure was simultaneously porous (28.9 vol% total porosity measured by FIB-SEM tomography, with 94.1% connected pores) and nanostructured, with mean ligament sizes of approximately 0.97 μm—a direct consequence of the Zener-like pinning of grain boundary migration by the evolving pore network during synthesis at temperatures below 0.75T_m .
Nitrogen interstitial alloying was confirmed at the atomic scale by APT measurements, which revealed approximately 1.8 at% N distributed within the martensite with evidence of local nitrogen clustering at planar defect sites—an observation that quantitatively substantiates the interstitial alloying step predicted by the second thermodynamic treasure map . The decisive role of NH₃ versus H₂ as the reducing atmosphere was further demonstrated by a comparative experiment: identical oxide pellets reduced under pure H₂, otherwise under identical conditions, yielded a ferrite-dominated microstructure with no martensite, confirming that the nitrogen from NH₃ is indispensable for driving the austenite–martensite transformation through its effect on the martensite start temperature . The specific Vickers hardness (HV normalized by density) of the porous martensitic alloy produced by fast cooling reached values exceeding those of a bulk Fe–10 at% Ni reference alloy produced by conventional melting–casting–recrystallization, highlighting the potential mechanical advantage of the nanostructured porous architecture .
Iron making is the biggest single cause of global warming. The reduction of iron ores with carbon generates about 7% of the global carbon dioxide emissions to produce ≈1.85 billion tons of steel per year. This dramatic scenario fuels eff orts to re-invent this sector by using renewable and carbon-free reductants and electricity. Here, the authors show how to make sustainable steel by reducing solid iron oxides with hydrogen released from ammonia. Ammonia is an annually 180 million ton traded chemical energy carrier, with established transcontinental logistics and low liquefaction costs. It can be synthesized with green hydrogen and release hydrogen again through the reduction reaction. This advantage connects it with green iron making, for replacing fossil reductants. the authors show t
Advanced Science - 2023 - Ma - Reducing [...]
PDF-Dokument [2.1 MB]
Synthesis Kinetics, Process Design, and Industrialization Pathways
The kinetic complexity of ADR and reactive dealloying–alloying demands careful attention in any upscaling strategy. Wei et al. proposed an Ashby-type kinetic conception map that demarcates distinct microstructural outcome regimes as a function of the P(NH₃)/P(H₂) ratio and temperature . At low heating rates and high ammonia partial pressures, the synthesis window allows concurrent tuning of porosity, grain size, and nitrogen content through the competing rates of pore creation (redox-driven) and pore annihilation (surface-energy-driven densification). Beyond the temperature at which all nitrides decompose, microstructural coarsening dominates and the nanostructured state is irreversibly lost. Below the critical dealloying temperature, incomplete oxygen removal results in retained oxide phases. The practical process window therefore lies between these two extremes, and its breadth is a strong function of heating rate and gas composition .
Jovičević-Klug et al. similarly highlighted that the process conditions—temperature, holding time, and atmosphere switching—determine not only the metallization degree but also the depth distribution and phase speciation of nitrogen within the pellet . Cross-sectional XRD line scans at 500 °C revealed pronounced shell–core heterogeneity, with nitrogen-rich nitride shells and oxide-containing cores, whereas at 700 °C, near-complete reduction was achieved across the pellet cross section, with nitriding concentrated at the outer surface and activated primarily during the cooling excursion . The lattice parameter expansion of α-Fe with increasing nitrogen content—measurable via systematic shifts in the {101} XRD reflection—provides a non-destructive proxy for the dissolved nitrogen level in the metallic fraction, enabling in situ or post-process diagnostics without destructive chemical analysis . Reoxidation experiments further demonstrated that the nitrogen-passivated ADR product showed superior resistance compared with the HyDR product when immersed in 0.1 mol NaCl aqueous solution, an observation that links directly to the passivating role of the Fe₄N surface layer identified by Ma et al. .
jovičević-klug-et-al-2024-thermal-kineti[...]
PDF-Dokument [3.8 MB]
Implications and Outlook for using Ammonia in Sustainable Metallurgy
Taken together, these three studies delineate a coherent and expanding research landscape in which ammonia transitions from a fossil-intensive industrial chemical to a cornerstone reagent of sustainable metallurgy. At the near-term, process engineering level, ADR represents a credible drop-in alternative to HyDR for solid-state ore reduction, compatible with existing shaft furnace geometries and downstream electric arc furnace steelmaking, with the added benefit of native reoxidation protection that reduces the logistical burden associated with shipping reactive DRI . At the intermediate process engineering level, the temperature and atmosphere management insights developed by Jovičević-Klug et al. provide the quantitative foundation—reduction kinetics, phase speciation maps, and lattice parameter diagnostics—needed to design industrial protocols that deliver a predictable and controllable nitrogen profile in the DRI product. At the frontier of physical metallurgy, the reactive vapor-phase dealloying–alloying framework of Wei et al. reveals that the conceptual boundaries between extraction metallurgy and alloy design are not fixed. By recognizing that the constituent atoms of the reacting gas are themselves potential alloying elements, one arrives at a synthesis philosophy in which a mixed oxide feedstock—potentially including metallurgical wastes or ore concentrates—is transformed in a single solid-state operation into a nanostructured, phase-engineered alloy with zero carbon footprint. Extensions of this paradigm to other alloy systems (Fe–Co–N permanent magnets, Fe–Cr–N corrosion-resistant alloys, metallic foams from oxide-contaminated scrap) were explicitly identified as immediately accessible targets . As green ammonia production costs continue to fall, driven by renewable electrolysis and improving Haber–Bosch efficiency, the economic and environmental logic of this dual-function reductant and alloying reagent will only strengthen—positioning ADR and its derivative alloy design concepts among the most consequential developments in twenty-first-century sustainable metallurgy.
For millennia, alloying has been the greatest gift from metallurgy to humankind: a process of mixing elements, propelling our society from the Bronze Age to the Space Age. Dealloying, by contrast, acts like a penalty: a corrosive counteracting process of selectively removing elements from alloys or compounds, degrading their structural integrity over time. We show that when these two opposite metallurgical processes meet in a reactive vapor environment, profound sustainable alloy design opportunities become accessible, enabling bulk nanostructured porous alloys directly from oxides, with zero carbon footprint. We introduce thermodynamically well-grounded treasure maps that turn the intuitive opposition between alloying and dealloying into harmony, facilitating a quantitative approach to na
sciadv.ads2140.pdf
PDF-Dokument [4.7 MB]