Stainless steels
Stainless steels are iron-base alloys containing at least 11 wt.% Chromium. They typcially contain less than 30 wt.% Cr and more than 50wt.% Fe. Stainless steels obtain their stainless characteristics because of the formation of an invisible and adherent chromium-rich oxide surface film. This oxide film has so few defects that oxygen cannot easily diffuse through it. The oxide establishes on the surface and heals itself in the presence of oxygen.
Some other alloying elements are often added to enhance specific characteristics. They include nickel, molybdenum, copper, titanium, aluminum, silicon, niobium, and nitrogen. Carbon is
usually present in amounts ranging from less than 0.03% to over 1.0% in certain martensitic grades (e.g. cutting steels have between 0.3 and 0.6 wt.% C). Corrosion resistance and mechanical
properties are commonly the principal factors in selecting a grade of stainless steel for a given application.
Stainless steels are commonly divided into five groups:
Martensitic stainless steels
Ferritic stainless steels
Austenitic stainless steels
Duplex (ferritic-austenitic) stainless steels
Precipitation-hardening stainless steels.
Martensitic stainless steels are essentially alloys of chromium and carbon that possess a martensitic crystal structure in the hardened condition. They are
ferromagnetic, hardenable by heat treatments, and are usually less resistant to corrosion than some other grades of stainless steel. Chromium content usually does not exceed 18wt.%, while
carbon content may exceed 1.0 wt.%. The chromium and carbon contents are adjusted to ensure a martensitic structure after hardening. Excess carbides may be present to enhance wear resistance
or as in the case of knife blades, to maintain cutting edges.
Ferritic stainless steels are chromium containing alloys with Ferritic, body centered cubic (bcc) crystal structures. Chromium content is typically less than 30%. The ferritic
stainless steels are ferromagnetic. They may have good ductility and formability, but high-temperature mechanical properties are relatively inferior to the austenitic stainless steels.
Toughness is limited at low temperatures and in heavy sections.
Austenitic stainless steels have a austenitic, face centered cubic (fcc) crystal structure. Austenite is formed through the generous use of austenitizing elements such as nickel,
manganese, and nitrogen. Austenitic stainless steels are effectively nonmagnetic in the annealed condition and can be hardened only by cold working. Some ferromagnetism may be noticed due
to cold working or welding. They typically have reasonable cryogenic and high temperature strength properties. Chromium content typically is in the range of 16 to 26wt.%; nickel content is
commonly less than 35wt.%.
Duplex stainless steels are a mixture of bcc ferrite and fcc austenite crystal structures. The percentage each phase is a dependent on the composition and heat treatment. Most Duplex
stainless steels are intended to contain around equal amounts of ferrite and austenite phases in the annealed condition. The primary alloying elements are chromium and nickel. Duplex stainless
steels generally have similar corrosion resistance to austenitic alloys except they typically have better stress corrosion cracking resistance. Duplex stainless steels also generally have
greater tensile and yield strengths, but poorer toughness than austenitic stainless steels.
Precipitation hardening stainless steels are chromium-nickel alloys. Precipitation-hardening stainless steels may be either austenitic or martensitic in the annealed condition.
In most cases, precipitation hardening stainless steels attain high strength by precipitation hardening of the martensitic structure.
Acta Materialia 165 (2019) 87 nanotwinne[...]
PDF-Dokument [7.1 MB]
Cracks often initiate at phase boundaries in conventional second phase reinforced alloys during cyclic loading, which limits their fatigue properties. Here, we prepared a nanotwin
strengthened 316L stainless steel consisting of nanotwinned and recrystallized grains by using plastic deformation and subsequent partial recrystallization annealing. Fatigue tests revealed
that interfaces separating hard nanotwinned grains from soft recrystallized ones exhibited excellent resistance to crack initiation. More than half of
the cracks (57% in number fraction) are found in recrystallized grains while a small fraction (11%) is observed at the interfaces between nanotwinned and recrystallized grains. This is ascribed
to the elastic homogeneity and cyclic deformation compatibility between nanotwinned and recrystallized grains.
Acta Materialia 81 (2014) 487 Nano-twin [...]
PDF-Dokument [2.1 MB]
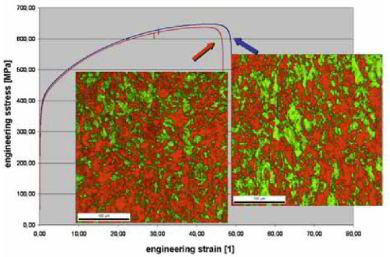
A novel type of duplex microstructure is generated in a single-phase austenitic steel (AISI 316L; X2CrNiMo19-12), consisting of plastically compliant recrystallized austenitic grains as the matrix containing coarse non-recrystallized grains with a nanotwinned austenitic (nt-c) structure as strengthening inclusions. This novel type of single-phase yet duplex microstructured steel exhibits an excellent combination of strength and ductility. We study the plastic co-deformation mechanisms between the nanotwinned and the recrystallized grains under tension using electron backscatter diffraction (EBSD) and transmission electron microscopy (TEM). At tensile strains below 5%, the nt-c grains nearly deform homogeneously in conjunction with the surrounding statically recrystallized (SRX) grains without generating notable strain localization near their interfaces.
Acta Materialia 97 (2015) 305-315
Y.F. Shen, N. Jia, Y.D. Wang, X. Sun, L. Zuo, D. Raabe
Suppression of twinning and phase transformation in an ultrafine grained 2 GPa strong metastable austenitic steel: Experiment and simulation
Acta Materialia 97 (2015) 305 UFG stainl[...]
PDF-Dokument [2.1 MB]
An ultrafine-grained 304 austenitic 18 wt.% Cr–8 wt.% Ni stainless steel with a grain size of 270 nm was synthesized by accumulative rolling (67% total reduction) and annealing (550 C, 150
s). Uniaxial tensile testing at room temperature reveals an extremely high yield strength of 1890 ± 50 MPa and a tensile strength of 2050 ± 30 MPa, while the elongation reaches 6 ± 1%.
Experimental characterization on samples with different grain sizes between 270 nm and 35 um indicates that both, deformation twinning and martensitic phase transformation are significantly
retarded with increasing grain refinement. A crystal plasticity finite element model incorporating a constitutive law reflecting the grain size-controlled dislocation slip and deformation
twinning captures the micromechanical behavior of the steels with different grain sizes. Comparison of simulation and experiment shows that the deformation of ultrafine-grained 304 steels
is dominated by the slip of partial dislocations, whereas for coarse-grained
steels dislocation slip, twinning and martensite formation jointly contribute to the shape change.
In-situ oberservation of the TRIP effect: martensite phase transformation in metastable austenitic stainless steel
Dislocations martensite transformation i[...]
PDF-Dokument [1.6 MB]
C. Herrera, D. Ponge, D. Raabe, steel research int. 79 (2008) No.6, page 482, Characterization of the Microstructure, Crystallographic Texture and Segregation of an As-cast Duplex Stainless Steel Slab
Herrera et al - steel research int. 79 ([...]
PDF-Dokument [1.4 MB]
Duplex stainless steels (DSSs) are based on the Fe-Cr-Ni system and consist of ferrite (30-70%) and austenite. DSSs have shown an excellent combination of resistance to general and localized
corrosion, stress corrosion cracking, high strength and low cost due to reduced contents of Ni and Mo [1-3]. DSSs are used in oil, gas, paper, desalination and petrochemical industries.
The main process steps in the industrial manufacturing of duplex stainless steel sheets are continuous casting, slab reheating, hot rolling, coiling, hot band heat treatment, cold rolling and final
recrystallization annealing. Particular attention during manufacturing of these steels has to be paid to the forming steps at high temperatures. Hot working of steels with two phases may cause
complications for several reasons: the precipitation of detrimental phases [1,4], such as σ-phase and M23C6, the low hot ductility, and edge crack formation. The ductility depends on different
factors like temperature, strain rate, microstructure and chemical composition. Moreover, it is affected by the different softening mechanisms in ferrite and austenite [5-9].
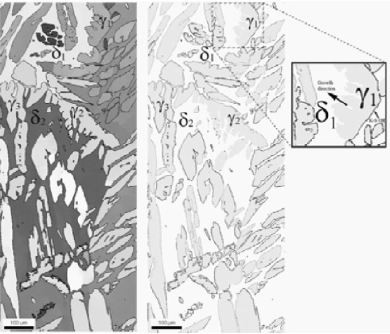
The formation of as-cast microstructures of duplex stainless steels depends on undercooling, cooling rates and subsequent solid state transformations which are influenced by the local chemical
composition [1,10-12]. Duplex stainless steels solidify by forming primary ferrite with austenite precipitates either from the liquid or in the solid state during cooling. The amount of austenite and
its morphology depend on the cooling rate. At increased cooling rate the amount of austenite is reduced [10-14].
The austenite can have a specific orientation relationship with the ferrite matrix. Grains that have been formed by a α-γ phase transformation should show a Bain, Nishiyama-Wassermann (N-W), or
Kurdjumov-Sachs (K-S) orienta-tion relationship. These orientation relationships can be described as a rotation of an angle ω about a common crystallographic axis (axis of rotation), this angle and
axis are known as angle/axis pair, Table 1.
Owing to these aspects associated with the microstructural state of the two phases prior to hot working the present study investigates in detail the microstructure, the crystallographic microtexture,
and the segregation of an as-cast duplex stainless steel slab produced by continuous casting (duplex stainless steel 1.4362).
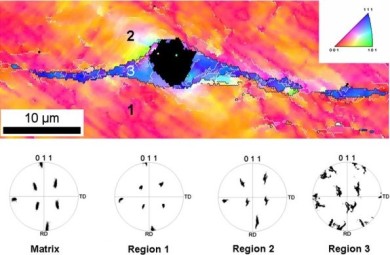
Here, the microstructure of a duplex stainless steel slab 1.4362 produced by continuous casting has been investigated by optical microscopy, scanning electron microscopy, EBSD and EDS. The slab showed different macrostructures through the thickness. The macrostructure can be divided into 3 types: fine equiaxed, columnar and coarse equiaxed grains. In all three regions, the volume fraction of each phase austenite and delta-ferrite is close to 50% and the hardness is very similar for both. The austenite has Kurdjumov-Sachs or Nishiyama-Wassermann relationship with the delta-ferrite. The slab does not show a strong segregation profile through the thickness. The delta-ferrite is enriched in Cr and Mo, while austenite is enriched in Ni and Mn.
Particle-stimulated nucleation (PSN) is investigated in Nb-containing ferritic stainless steel. Coarse-grained sheets were cold rolled to 80 pct thickness reduction and annealed from 973 K to 998 K (7
PSN-ferritic-steels-2013-METALL-TRANS-A-[...]
PDF-Dokument [1.2 MB]
Acta Materialia 59 (2011) 4653-4664, C. Herrera, D. Ponge, D. Raabe, Design of a novel Mn-based 1 GPa duplex stainless TRIP steel with 60% ductility by a reduction of austenite stability
Herrera Ponge Raabe Acta Materialia 59 ([...]
PDF-Dokument [1.0 MB]
Duplex stainless steels (DSS) which consist of ferrite and austenite (potentially plus corresponding displacive transformation products) are characterized by an excellent
combination of good mechanical properties and corrosion resistance. The new alloy class proposed in this work uses Mn, C, and N (instead of only Ni) to stabilize the
austenite (Fe–19.9Cr–0.42Ni–0.16N–4.79Mn–0.11C–0.46Cu–0.35Si, wt.%) [1–5]. Our design strategy aims at an alloying corridor to obtain optimum austenite stability. The
motivation for that is twofold. First, the austenite should be sufficiently unstable that a transformation-induced plasticity (TRIP) effect is initiated upon loading. Second, the
austenite should be sufficiently stable that the TRIP effect occurs over a wide strain regime, specifically at high strains, where strain hardening reserves are usually more desirable
than at
low strains. The TRIP mechanism is based on deformation-stimulated displacive transformation of metastable austenite (face centered cubic Fe–Mn–N–C phase) to martensite (metastable or body
centered orthorhombic or hexagonal phase) and the matrix plus martensite plasticity required to accommodate the transformation misfit. In the new alloy concept Mn not only replaces Ni
but
also increases the solubility of interstitial elements such as C and N, which promote austenite stabilization and provide additional solid solution strengthening [6]. N also enhances
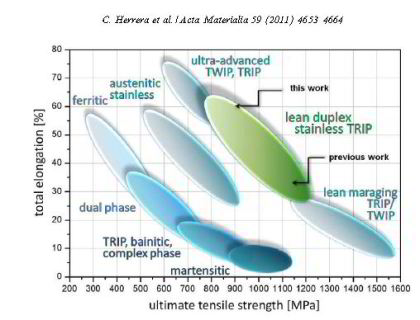
the resistance to localized corrosion and delays sensitization [7,8]. Commercial grade lean DSS (LDX 2101 and 1.4362,
Fe–22Cr–4Ni–0.3N–0.03C–0.3Mo, wt.%) exhibit a total elongation of up to 30% and ultimate strength levels of up to 600–700 MPa (Fig. 1). As well as these alloys with their limited
ductility some studies have addressed improved lean DSS alloys [3,4]. Wang et al. [3] studied a new group of Ni-free, high strength and corrosion resistant DSS. These Ni-free alloys
exhibited yield and ultimate
tensile strengths of about 550 and 750 MPa, respectively, at an elongation to fracture of 40%. The materials exhibited excellent resistance to stress corrosion cracking (SCC)
in chloride solutions. Wessman et al. [4] studied the effects
of the replacement of Ni by Mn and N on the mechanical and corrosion properties in DSS alloys with up to 3 wt.% Mo. They found that the pitting corrosion resistance for the low and
Ni-free alloys decreased while the yield stress and tensile stress increased with higher N andMn contents. The effect of Ni on the mechanical properties was not significant.
Here, we report on the microstructure, texture and deformation mechanisms of a novel ductile lean duplex stainless steel (Fe–19.9Cr–0.42Ni–0.16N–4.79Mn–0.11C–0.46Cu–0.35Si, wt.%). The austenite is
stabilized by Mn, C, and N (instead of Ni). The microstructure is characterized by electron channeling contrast imaging (ECCI) for dislocation mapping and electron backscattering diffraction
(EBSD) for texture and phase mapping. The material has 1 GPa ultimate tensile strength and an elongation to fracture of above 60%. The
mechanical behavior is interpreted in terms of the strength of both the starting phases, austenite and ferrite, and the amount, dispersion, and transformation kinetics of the mechanically
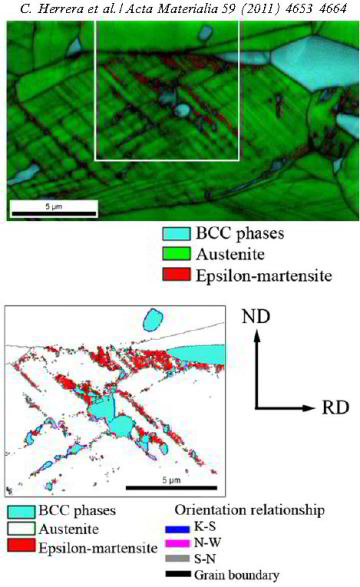
induced martensite (TRIP effect). Transformation proceeds from austenite to hexagonal martensite to near cubic martensite). The epsilon-martensite forms in the austenite with an orientation
relationship close to Shoji-Nishiyama. The alpha-martensite nucleates at the intersections of deformation bands, especially e-bands, with Kurdjumov–Sachs and Nishiyama–Wassermann relationships.
The ferrite deforms by dislocation slip and contains cell substructures.
Acta Materialia 59 (2011) 4653
Design of a novel Mn-based 1 GPa duplex stainless TRIP steel with 60% ductility by a reduction of austenite stability
C. Herrera, D. Ponge, D. Raabe
Acta Materialia 59 (2011) 4653 Cr Mn dup[...]
PDF-Dokument [1.0 MB]
Here we report on the microstructure, texture and deformation mechanisms of a novel ductile lean duplex stainless steel (Fe–19.9Cr–0.42Ni–0.16N–4.79Mn–0.11C–0.46Cu–0.35Si, wt.%). The austenite is stabilized by Mn, C, and N (instead of Ni). The microstructure is characterized by electron channeling contrast imaging (ECCI) for dislocation mapping and electron backscattering diffraction (EBSD) for texture and phase mapping. The material has 1 GPa ultimate tensile strength and an elongation to fracture of above 60%. The mechanical behavior is interpreted in terms of the strength of both the starting phases, austenite and ferrite, and the amount, dispersion, and transformation kinetics of the mechanically induced martensite (TRIP effect).
steel research int. 79 (2008) No. 6 pages 440 ff
Advances in the Optimization of Thin Strip Cast Austenitic 304 Stainless Steel
D. Raabe, R. Degenhardt, R. Sellger, W. Klos, M. Sachtleber, L. Ernenputsch
steel research int. 79 (2008) 440 strip [...]
PDF-Dokument [788.8 KB]
Stainless steel strips are conventionally manufactured by continuous casting, slab reheating, hot rolling, hot strip annealing, and pickling. Thin strip steel casters offer a competitive
efficient alternative to industrially produce such steels when compared to the conventional thick-slab production lines. Today’s advanced twin roll thin strip casters for the production of stainless
steels combine the two operations of casting liquid metal between two rolls and subsequently imposing in-line hot deformation steps to produce hot rolled thin strips that are directly coilable [1-5]
(Figure 1). Twin roll thin strip casting of steels thus eliminates major steps required in conventional production, for instance slab handling, slab reheating, hot strip roughening / break-down
reversing rolling, and hot rolling in a conventional hot rolling mill (set of 5-7 four-high stands) (Figure 2). Both thin strip cast material and conventionally produced hot strips can be further
cold rolled and recrystallization annealed, depending on the desired final thickness and properties.
Thin strip casting of stainless austenitic steel (AISI 304 / 1.4301 in this work) provides a number of significant advantages in comparison to the conventional slab processing method. First, the thin
strip casting method permits the entire continuous conventional hot rolling process to be bypassed. The thin strip casting method is even capable of producing strips with a smaller thickness than
conventional production routes. At the same time it allows one to produce steel strips which are difficult to be cast or hot rolled by conventional methods, such as for instance some complex highly
alloyed austenitic grades. Second, it offers a significantly higher solidification rate, which leads to microstructures with reduced dendrite arm spacing, reduced microsegregation, and smaller
inclusion sizes when compared to conventional slabs. Third, the thin strip route allows exploiting higher and locally different heat fluxes which may lead to alternative solidification modes with
respect to the primary γ / δ phase ratio close to the strip of the surface. Fourth, the weak crystallographic texture and low through-thickness texture gradients of strip cast steels, which
predetermine the forming properties and strength of the final strip, result in reduced anisotropy when compared to conventional hot strip material [6-13].Fifth, the microstructure and properties of
strip cast austenitic 304 stainless steels are equivalent to those obtained by conventional processing. Also, strip cast austenitic stainless steels often reveal a more homogeneous microstructure
through the strip thickness than conventional hot rolled material. This means that the scatter in the mechanical properties may be even lower than in the case of conventionally produced stainless
steels that are known to reveal through-thickness gradients of their texture and microstructure [14-19]. Finally, the strip casting route is the most environmentally friendly, energy saving, and
CO2-sensitive way to produce steel strips.
In this study we present the latest advances in the optimization of the microstructure and properties of strip cast austenitic stainless steels (AISI 304, 1.4301). We discuss in part the influence of
some of the relevant process parameters (e.g. coiling temperature, in-line hot rolling temperature, lubrication, in-line rolling speed, heat treatments) for optimizing the microstructure and
properties of the final strips.
More specific this study is about the latest advances in the optimization of the microstructure and properties of thin strip cast austenitic stainless steel (AISI 304, 1.4301). Concerning the processing steps the relevance of different thin strip casting parameters, in-line forming operations, and heat treatments for optimizing microstructure and properties have been studied. The microstructures obtained from the different processing strategies were analysed with respect to phase and grain structures including the grain boundary character distributions via EBSD microtexture measurements, the evolution of deformation-induced martensite, the relationship between delta ferrite and martensite formation in austenite, and the texture evolution during in-line deformation. It is observed that different process parameters lead to markedly different microstructures and profound differences in strip homogeneity. It is demonstrated that the properties of strip cast and in-line hot rolled austenitic stainless steels are competitive to those obtained by conventional continuous casting and hot rolling. This means that the thin strip casting technique is not only competitive to conventional routes with respect to the properties of the material but also represents the most environmentally friendly, flexible, energy-saving, and modern industrial technique to produce stainless steel strips.
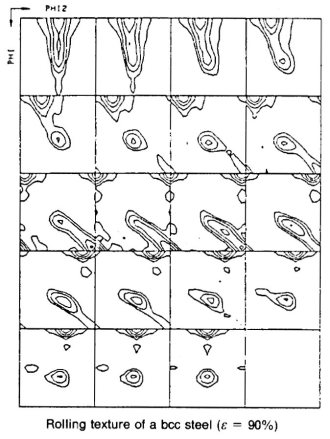
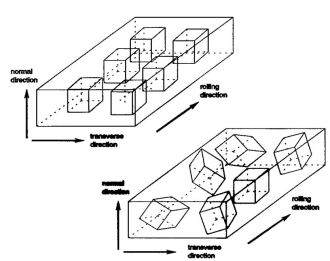
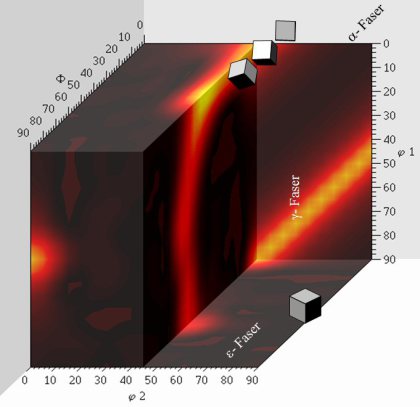
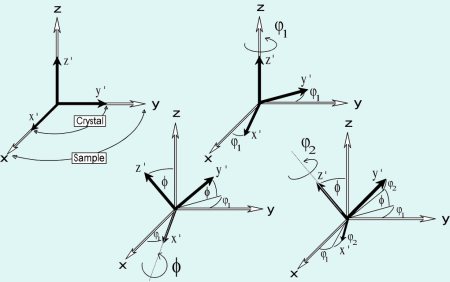
Rolling and recrystallization textures of BCC steels
Martin Holscher, Dierk Raabe and Kurt Lucke
steel research 62 (1991) No. 12 page 567
steel research 62 (1991) No. 12 page 567[...]
PDF-Dokument [748.4 KB]
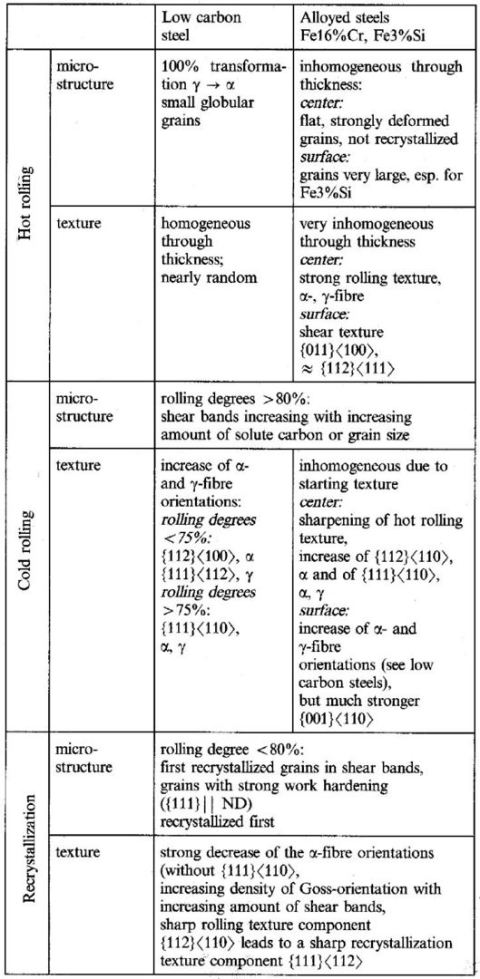
The rolling and recrystallization textures of the different types of bcc steels often show great similarities, but also exhibit characteristic
differences which, e.g. depend upon starting texture, microstructure and condition of precipitations.
In this paper this behaviour will be discussed for three examples belonging to three entirely different types of steels.
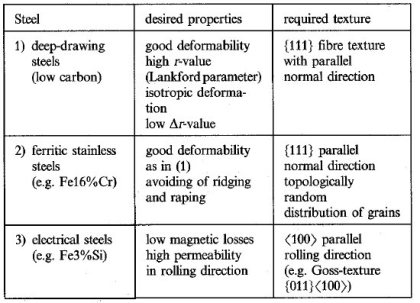
Deep-drawing steels (e.g, low-carbon steel). Here the most important property is a good deformability in deep drawing. This behaviour is favoured by a texture formed in such a way that during deepdrawing the material flow occurs from the width and not from the thickness of the sheet and that it is equal for different directions in the sheet plane, i.e. by materials with the high r-value and a low !J.rvalue (r is the Lankfort parameter). Both properties can be achieved by a texture which after recrystallization consists of a homogeneous strong fibre texture with a {111} plane parallel to the sheet plane.
Ferritic stainless steels (e.g. Fe16%Cr). Here again the requirements for deep drawability should be fulfilled, but additionally the ridging which often occurs in
Cr-steels should be suppressed. This again means a {111} fibre texture but also a topologically random arrangement of crystallites should be achieved'),
Electrical steels (e.g, Fe3%Si). For the use of these steels in transformers high magnetic permeability for magnetisation in rolling direction is required which can be obtained by a <100) direction parallel to the rolling direction. Technologically this can be achieved by forming a very sharp Goss texture {011}<100) by secondary recrystallization.