Why study interface segregation in alloys atomic scale?
Advanced metallic alloys for structural and functional applications can be manipulated down to the nanometer scale, for instance through heat treatments. This applies particularly to interfaces in high strength alloys: Their structural and chemical state can be a decisive factor whether the bulk material reacts brittle or ductile against external loads. In this context segregation of solute atoms to grain boundaries plays a major role. This webpage gives a little introduction to the current state of chemical grain boundary analysis in advanced alloys by use of atom probe tomography. The necessity and challenge in coupling such near-atomic chemical interface characterization via atom probe tomography with structural analysis via electron microscopy of the same material region is discussed and corresponding examples are presented. Theoretical analysis of grain boundary segregation phenomena is conducted on the basis of the Gibbs and McLean isotherm approaches and also in terms of phase field approaches.
Introduction to the Segregation Engineering Principle
The term segregation engineering (SE) describes a self-organized microstructure manipulation method that is based on the combination of (i) confined thermodynamically driven chemical Gibbs segregation via solute decoration to specific lattice defect traps, and (ii) the associated structural, order or phase changes triggered at these decorated sites. The concept of SE works particularly well for steels and Ti alloys owing to their rich spectrum of available allotropic phase transformations. Below, we discuss a number of cases where local solute segregation, and the associated ordering phenomena at lattice defects, lead to confined martensite-to-austenite reversion in Fe-Mn and Fe-C steels but also in Ti alloys.
How does Segregation Engineering work at Lattice Defects in Metals ?
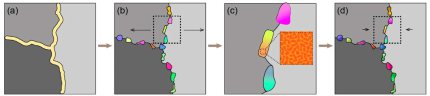
In this case the targeted segregation engineering effects occur in a spatially confined manner, namely, at lattice defects, which act as efficient and highly thermodynamically attractive solute traps, thus, entailing strong solute segregation when heat treated into regimes that allow for sufficient solute diffusion. The resulting combinations of local strain fields at such lattice defects on the one hand, and the locally altered chemical composition at these defects on the other hand, can initiate structural transitions, solute ordering, and phase transformation states in such cases where the sample is heated into a phase regime which is intercritical at the decorated and distorted defect region. When aiming at the local modification of the microstructure, this SE approach is applied in such a way that these critical conditions are only fulfilled at the lattice defect regions, leaving the adjacent matrix essentially unaffected. These effects, which are essentially built on the decoration driving forces phenomenologically described by the Gibbs adsorption isotherm, are referred to as equilibrium segregation engineering.
Grain and phase boundary segregation in high strength steels
Strong, damage-tolerant, and functional steels are the backbone for multiple innovations in the fields of energy, transportation, health, safety, machinery, and industrial infrastructure. Examples are advanced Fe-Cr steels for high-temperature applications in emission-reduced turbines; weight reduced and ultra high strength Fe-Mn steels for light-weight and yet passenger-protecting mobility; soft magnetic Fe-Si steels, semi-amorphous steels, and iron-based metallic glasses for low-loss electrical motors and wind energy generators; radiation resistant steels for nuclear and fusion power plants; or Fe-Cr-Ni stainless steel tubes for direct solar thermal power plants.
The fact that steels are often 'hidden' behind the final product sometimes conceals the truth that without innovation in this field many advanced industrial products would be impossible to realize. Awareness of the profound recent advances in steel research sometimes suffers a bit from its engineering success: Although steels are key components in many zero-damage-tolerance safety design parts (planes, trains, cars, bridges, wind mill turbines, power plants, etc.) they very rarely fail, hence, awareness of the high relevance of steels is not as ubiquitous as the products that are made of it. This is surprising when considering that the annual steel production exceeds 1.8 Billion tons and that most high-performance steels are complex nanostructured materials.
The applications mentioned above require the development of improved high strength and yet ductile and damage tolerant steels. Most traditional hardening mechanisms, however, such as solute solution, dislocation multiplication, or precipitation, albeit leading to high strength, often may cause a decrease in ductility, i.e., rendering the material brittle and susceptible for failure. This phenomenon is sometimes referred to as the inverse strength-ductility problem.
In this context the average grain size and the nature of the grain boundaries involved in terms of their thermodynamic, kinetic and structural characteristics are important factors. Better understanding the role of grain boundaries plays particularly an essential role for designing and manufacturing nanostructured steels with higher ductility. Here I refer specifically to grain boundaries as defects in advanced high strength steels and their susceptibility to undergo chemical decoration by attracting solute atoms. This phenomenon, also referred to as grain boundary segregation, is characterized by the inhomogeneous distribution of solute atoms between the grain boundary and the abutting grains. When interpreting the interface as a separate phase this effect resembles a partitioning phenomenon as observed among abutting phases. The concentration of the solutes on the grain boundary exceeds their solubility in the grain interior, sometimes by a factor of 2-3 and sometimes even by up to several orders of magnitude. A rough approximation for the segregation tendency of a solute is its bulk solubility, namely, the smaller the bulk solubility, the higher is the enrichment factor of that element on the interface. Naturally, exceptions may apply from this simple rule.
The topic of grain boundary segregation is of high relevance in the context of alloy design, because the damage tolerance of structural materials, such as steels and Ti alloys, is often limited by segregation at grain boundaries. This phenomenon can hence profoundly affect the structure and the mechanical properties of advanced materials: The overall resistance of steels against damage and failure depends on two main material properties. Firstly, steels require during plastic straining a high, if possible, permanent capability to undergo additional deformation stimulated strain hardening. This property of the material, e.g. such as that realized in TRIP (transformation induced plasticity), TWIP (twinning induced plasticity), and TRIPLEX (weight-reduced, austenitic or austenitic-ferritic with kappa carbides) steels, ensures that any tendency towards macroscopic and microstructural localization associated with geometrical softening during deformation is compensated for by strain hardening. This makes the material locally stronger and hence resistant to the further progress of plastic localization zones that might otherwise become fatal to the material. Secondly, steels require mechanisms that suppress the formation and specifically the growth of cracks. While tiny flaws, i.e. initial crack nuclei, are always present in bulk materials, specific attention and understanding has to be placed on mechanisms that help avoiding, branching, deflecting, or even closing cracks that are capable of growing or already started to grow.
In the latter context it has been often observed that in high strength steels it is particularly the internal interfaces that are relatively weak against crack formation and growth. This is
due to the fact that elementary crack growth proceeds by debonding and dislocation emission at the crack tip, i.e. by decohesion of the atomic bonds and the ability of the material to create a
plastic relaxation zone around the tip of a proceeding crack tip. Due to the reduced atomic stacking and hence smaller lattice coherence in the grain boundary zones compared to the bulk grain
interiors, cracks can in high strength steels often proceed more easily along grain boundaries than through the grain interior. This applies particularly to plastically strained steels, often
associated with strain path changes, when the intrinsic strain hardening capacity of the material is gradually exhausted.
How does solute atomic segregation to internal interfaces in steels matter in such a scenario? When grain boundaries are affected by equilibrium segregation, either via inherited segregated solutes
from a preceding high temperature processing step (i.e. decoration of former austenite grain boundaries) or upon modest tempering after quenching (i.e. decoration of martensite grain boundaries), the
structure and the atomic bonds change within the decorated grain boundary. Here, different scenarios are conceivable: Firstly, segregation of certain atomic species might enhance the
grain boundary structure, coherence, and bonding (grain boundary strengthening). Secondly, the reverse might be true, namely, the further loss of coherence and electronic bonding (interface
weakening) at grain boundaries due to segregation. Thirdly, segregation could be strong enough to finally result in a phase transformation of the grain boundary region or support the formation
of second phases (grain boundary precipitation, complexions, phase formation on grain boundaries, phase transformation of grain boundaries).
Besides these possible direct mechanical and structural changes in the intrinsic grain boundary properties due to equilibrium segregation, also the grain boundary energy is affected. More
precisely, the reduction in grain boundary energy provides the driving force behind many of these phenomena. As will be outlined in more detail below the original formulation of the Gibbs adsorption
isotherm and also the McLean variant of this concept, when being adopted to segregation at internal interfaces, describes the reduction in the total system free energy as the driving force for
equilibrium segregation phenomena. Since the bulk grain depletion and the associated bulk free energy change is negligible in this context, the main thermodynamic driving force for segregating
solutes to grain boundaries lies in the reduction of the interface free energy. Kirchheim generalized this concept to the defectant theory, describing the general phenomenon of solute segregation to
lattice defects with the result that their free energy is reduced. In a thought experiment he shows that in extreme cases the defect free energy could even become smaller than that of the bulk phase
surrounding it, meaning that the defects would be stabilized against the defect-free bulk phase. This case will not be considered further here though.
The energy reduction due to solute segregation also plays an indirect role for the relationship between segregation and ductility because the lower grain boundary energy reduces the driving force for
competitive capillary driven grain coarsening (grain growth). Hence, materials with smaller grain boundary energy can, under the same thermomechanical treatment as applied to materials without
grain boundary segregation, have a smaller and at the same time rather stable average grain size. This effect could also improve both the strength and the ductility of the steel via the
Hall-Petch effect provided that the specific type of segregation does not lead to interface embrittlement.
When properly understood and quantitatively characterized, these various interconnected effects between grain boundaries and the associated equilibrium segregation effects in high strength steels may
open novel pathways to nanoscale engineering of damage tolerant high strength steels.
Does Segregation Engineering also work with Scheil segregation?
Alternatively to equilibrium Gibbs segregation effects, it is also conceivable to exploit such decoration and confined manipulation / transformation effects for the case of non-equilibrium
compositional gradients that are inherited either from a Scheil-Gulliver type solidification inhomogeneity or from incomplete intercritical phase dissolution (incomplete vessel phase dissolution).
Such scenarios, which are based on kinetically acquired compositional inhomogeneities, are referred to as non-equilibrium segregation engineering.
When applying equilibrium SE specifically to grain boundaries we refer to this type of interface manipulation via solute decoration in conjunction with confined ordering or even transformation as
'grain boundary segregation engineering'.
Self-organized Microstructures through Segregation Engineering
For creating self-organized microstructures by the segregation engineering approach one has to take certain thermodynamic and kinetic rules into account. The thermodynamic rules include the requirement to consider also the competition with the formation of alternative phases in the bulk matrix (rather than being confined to specific defects), the consideration of the equilibrium or non- equilibrium segregation coefficients, co-segregation effects, phase stability (relevant when designing confined states that can undergo spatially confined athermal phase transformations), as well as the interplay of chemical and mechanical driving forces. The kinetic rules include consideration of the diffusion coefficients as well as of thermal versus diffusional phase transformation pathways.
The main idea behind the segregation engineering concept lies in the approach to consider solute decoration to defects not just as an undesired side effect associated with processing and heat treatment but to exploit it as a tool for the site-specific manipulation of interfaces with the aim to stabilize small grain sizes or to create intrinsically organized nano- or microstructures consisting of the initial matrix phase and segregation-induced ordered or transformation phase regions that can be rendered to follow the template of the landscape of the lattice defect structure.
Acta-Mater-Nano-Austenite-reversion-2013[...]
PDF-Dokument [1.1 MB]
Acta 2019 Thermodynamics of grain bounda[...]
PDF-Dokument [4.0 MB]
What is Grain Boundary Segregation Engineering?
Grain boundaries are ubiquitous defects in metallic alloys governing a range of properties, such as tensile strength, fatigue resistance, fracture toughness, strain hardening, brittleness, conductivity, or corrosion.
Unlike bulk phases they are planar and require 5 kinematic parameters to be crystallographically characterized (crystallographic disorientation of the adjacent grains, plane inclination relatiuve to these grains). Hence, grain boundaries cannot be regarded as scalar objects with homogeneous properties but as a high dimensional class of defects. For example, even for highly coherent grain boundaries such as first order twins (Σ3 CSL (coincidence site lattice)) exponential property changes can occur if the grain boundary plane is misaligned by only a few degrees from its most coherent position.
Grain boundaries can either weaken (inter-crystalline fracture, stress corrosion cracking) or strengthen (Hall-Petch effect) polycrystalline metallic materials. In either case the types of grain boundaries involved affect the specific material response substantially as was discussed particularly in the context of highly coherent versus random grain boundaries. These observations led to the concept of grain boundary engineering which aims at designing polycrystals with specific grain boundary distributions.
The next logical step in this context lies in also considering and engineering the segregation and its effects on grain boundaries. This is an obvious thought since many if not all grain boundary properties are accessible to decoration-driven manipulation such as cohesion; energy; fracture resistance; electrical conductivity; transport coefficients; electrochemical properties; hydrogen embrittlement; mobility; and resistance to or sources of dislocations to name only a few effects that are relevant for structural alloys.
We refer to such manipulation of grain boundaries via solute decoration and even confined transformation as 'grain boundary segregation engineering' (GBSE) or just 'segregation engineering'. Applying GBSE needs to involve both, thermodynamic (segregation coefficient; co-segregation; competing bulk phases) and kinetic (diffusion) influence factors. Hence, not only the associated crystallographic degrees of freedom and all segregated elements should be considered but also time and temperature. However, as will be addressed below, GBSE can for a number of alloys even be applied when knowing the average segregation level to non-ordered or ‘random’ grain boundaries, i.e. a full 5D crystallographic description is not in all cases required.
How can grain boundary segregation be influenced ?
Grain boundary segregation changes with time when the heat treatment or application temperature is high enough for diffusion.
Hence, also the grain boundary properties may alter. GBSE considers solute decoration not as an undesired and inherited phenomenon but instead utilizes segregation as a tool for site-specific
manipulation that allows for optimizing specific grain boundary structures, compositions and properties that enable the design of beneficial material behavior. More specific, grain boundary
segregation can be used as a microstructure design method since solutes affect the structure, phase state and atomic bonds within the decorated interface. Different scenarios are conceivable: First,
segregation of certain solutes might enhance grain boundary cohesion (grain boundary strengthening). Second, the reverse might apply, namely, reduction in cohesion and bonding strength (interface
weakening) at grain boundaries due to segregation. Third, segregation could be strong enough, in conjunction with the avalable interfacial energy and local stresses, to result in a phase
transformation of or at the grain boundary region. Related to the latter point is the possibility for the formation of complexions, i.e. interface stabilized phases.
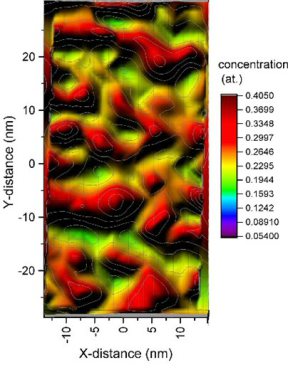
Elemental segregation to grain boundaries (GBs) can induce structural and chemical transitions at GBs along with significant changes in material properties. The presence of multiple principal elements interacting in high-entropy alloys (HEAs) creates multiple co-segregation effects.
Acta Materialia 2019 Segregation-driven [...]
PDF-Dokument [3.9 MB]
Does Segregation Engineering also work in High Entropy Alloys?
Elemental segregation to grain boundaries (GBs) can induce structural and chemical transitions at GBs along with significant changes in material properties. The presence of multiple principal
elements interacting in high-entropy alloys (HEAs) makes the GB segregation and interfacial phase transformation
a rather challenging subject to investigate. Here, we explored the temporal evolution of the chemistry for general high-angle GBs in a typical equiatomic FeMnNiCoCr HEA during aging heat
treatment through detailed atom probe tomography (APT) analysis. We found that the five principal elements segregate
heterogeneously at the GBs. More specifically, Ni and Mn co-segregate to some regions of the GBs along with the depletion of Fe, Co and Cr, while Cr is enriched in other regions of the GBs where
Ni and Mn are depleted. The redistribution of these elements on the GBs follow a periodic characteristic, spinodal-like compositional modulation. The accumulation of elements at the GBs can
create local compositions by shifting their state from a solid solution (like in the adjacent bulk region) into a spinodal regime to promote interfacial phase-like transitions as
segregation proceeds. These results not only shed light on phase precursor states and the associated nucleation mechanism at GBs in alloy systems with multiple principal elements but also
help to guide the microstructure design of advanced HEAs in which formation of embrittling phases at interfaces must be avoided.
How can grain boundary segregation be used to engineer microstructures ?
An important question in that context is the accessibility of these segregation-dependent grain boundary features via an 'engineering' approach. Indeed various examples are conceivable where such misorientation- and plane-sensitive design approach can work, namely, diffusion annealing, grain growth heat treatment, and local phase transformation effects.
A typical example for an engineering heat treatment approach to tune a desired segregation level at grain boundaries lies in a combined diffusion plus grain growth heat treatment: Since the reduction and grain boundary energy may depend on both, the grain boundary plane inclination and its crystallographic misorientation, such low energy configurations can be obtained for instance by a grain growth heat treatment, where low-energy interfaces prevail owing to their comparably small capillary driving force. Another broadly applicable example of grain boundary segregation engineering lies in the reduction of the average grain size due to an overall drop in grain boundary energy caused by equilibrium segregation.
Although GBSE can be applied to such diverse systems as solar cells, superconductors, or ceramics we focus here on recent developments pertaining to metallic alloys used for structural applications.
In these materials we see particularly high relevance and opportunities of the GBSE concept for designing improved ultra fine grained and nanostructured metallic alloys where grain boundaries constitute a large fraction of the material. Particularly alloys with nanoscaled grain size suffer often from very low ductility in conjunction with inter-crystalline damage. In such materials particularly the internal interfaces are prone to void formation and damage initiation.
Segregation engineering enables confined transformation effects at lattice defects in materials
The microstructure of complex steels can be manipulated by utilising the interaction between the local mechanical distortions associated with lattice defects, such as dislocations and grain boundaries, and solute components that segregate to them. Phenomenologically these phenomena can be interpreted in terms of the classical Gibbs adsorption isotherm, which states that the total system energy can be reduced by removing solute atoms from the bulk and segregating them at lattice defects. Here we show how this principle can be utilised through appropriate heat treatments not only to enrich lattice defects by solute atoms, but also to further change these decorated regions into confined ordered structural states or even to trigger localized decomposition and phase transformations. This principle, which is based on the interplay between the structure and mechanics of lattice defects on the one hand and the chemistry of the alloy's solute components on the other hand, is referred to as segregation engineering. In this concept solute decoration to specific microstructural traps, viz. lattice defects, is not taken as an undesired effect, but instead seen as a tool for manipulating specific lattice defect structures, compositions and properties that lead to beneficial material behavior. Owing to the fairly well established underlying thermodynamic and kinetic principles, such local decoration and transformation effects can be tuned to proceed in a self-organised manner by adjusting (i) the heat treatment temperatures for matching the desired trapping, transformation or reversion regimes, and (ii) the corresponding timescales for sufficient solute diffusion to the targeted defects. Here we show how this segregation engineering principle can be applied to design self-organized nano- and microstructures in complex steels for improving their mechanical properties.
Segregation Engineering to Reduce Thermal Conductivity of Thermoelectrics
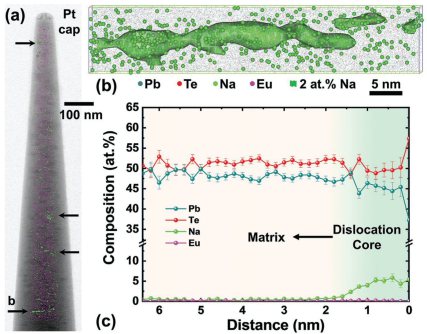
Dislocations play an important role in thermal transport by scattering phonons. Nevertheless, for materials with intrinsically low thermal conductivity, such as thermoelectrics, classical models require exceedingly high numbers of dislocations (>10^12 cm–2) to further impede thermal transport. In this work, a significant reduction in thermal conductivity of Na0.025Eu0.03Pb0.945Te is demonstrated at a moderate dislocation density of 1 × 10^10 cm–2. Further characteristics of dislocations, including their arrangement, orientation, and local chemistry are shown to be crucial to their phonon-scattering effect and are characterized by correlative microscopy techniques. Electron channeling contrast imaging reveals a uniform distribution of dislocations within individual grains, with parallel lines along four <111> directions. Transmission electron microscopy (TEM) shows the parallel networks are edge-type and share the same Burgers vectors within each group. Atom probe tomography reveals the enrichment of dopant Na at dislocation cores, forming Cottrell atmospheres. The dislocation network is demonstrated to be stable during in situ heating in the TEM. Using the Callaway transport model, it is demonstrated that both parallel arrangement of dislocations and Cottrell atmospheres make dislocations more efficient in phonon scattering. These two mechanisms provide new avenues to lower the thermal conductivity in materials for thermal-insulating applications.
Parallel Dislocation Networks Cottrell A[...]
PDF-Dokument [1.7 MB]
Interface Segregation Engineering in Additive Manufacturing
Can Segregation Engineering also serve in the design of more robust and crack-free microstructures in Additive Manufacturing? Yes. We were able to reduce hot tearing in additive manufacturing of an Al x CoCrFeNi high-entropy alloy by grain boundary segregation engineering.
One fundamental problem in alloy design for additive manufacturing (AM) is hot tearing. We tried to solve that by segregation engineering, with the specific aim to alter the residual stress states at the interdendritic and grain boundary regions and consequently prevent hot tearing. Here, in situ Al alloying is introduced into an existing hot-cracking susceptible high-entropy alloy CoCrFeNi. We find that within a certain range of compositions, such as Al 0.5 CoCrFeNi, the hot crack
density was drastically decreased. During the solidification of this specific alloy composition, Al is firstly ejected from the primary dendritic face-centred cubic (FCC) phase and segregates into the interdendritic regions. Spinodal decomposition then occurs in these Al-enriched regions to form the ordered B2 NiAl and disordered body-centred cubic (BCC) Cr phases. Due to the higher molar volume and lower homologous temperatures of these B2/BCC phases, the inherent residual strain is accommodated and transformed from a maximum 0.006 tensile strain in CoCrFeNi to a compressive strain of ~0.001 in Al 0.5 CoCrFeNi. We suggest that this interface segregation engineering method is new pathway to counteract hot tearing in additive manufacturing.
Acta Materialia 204 (2021) 116505
Acta Mater 2020 hot tearing grain bounda[...]
PDF-Dokument [5.9 MB]
SCIENCE, SEPTEMBER 2015 VOL 349 ISSUE 6252
Dislocations as Linear Complexions Scien[...]
PDF-Dokument [1.5 MB]
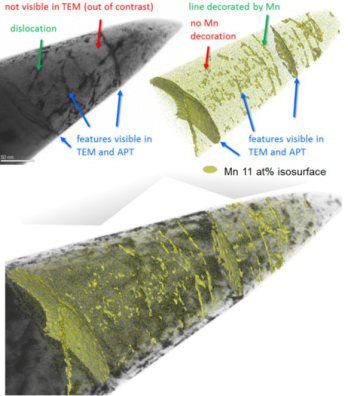
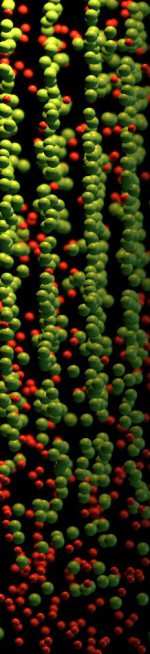
For 5000 years, metals have been mankind’s most essential materials owing to their ductility and strength. Linear defects called dislocations carry atomic shear steps, enabling their
formability. We report chemical and structural states confined at dislocations. In a body-centered cubic Fe–9 atomic percent Mn alloy, we found Mn segregation at dislocation cores during
heating, followed by formation of face-centered cubic regions but no further growth. The regions are in equilibrium with the matrix and remain confined to the dislocation cores with
coherent interfaces.The phenomenon resembles interface-stabilized
structural states called complexions. A cubic meter of strained alloy contains up to a light year of dislocation length, suggesting that linear complexions could provide opportunities to
nanostructure alloys via segregation and confined structural states.
Acta Materialia 86 (2015) 182-192
Acta Materialia 86 (2015) 182 Kuzmina Gr[...]
PDF-Dokument [1.8 MB]
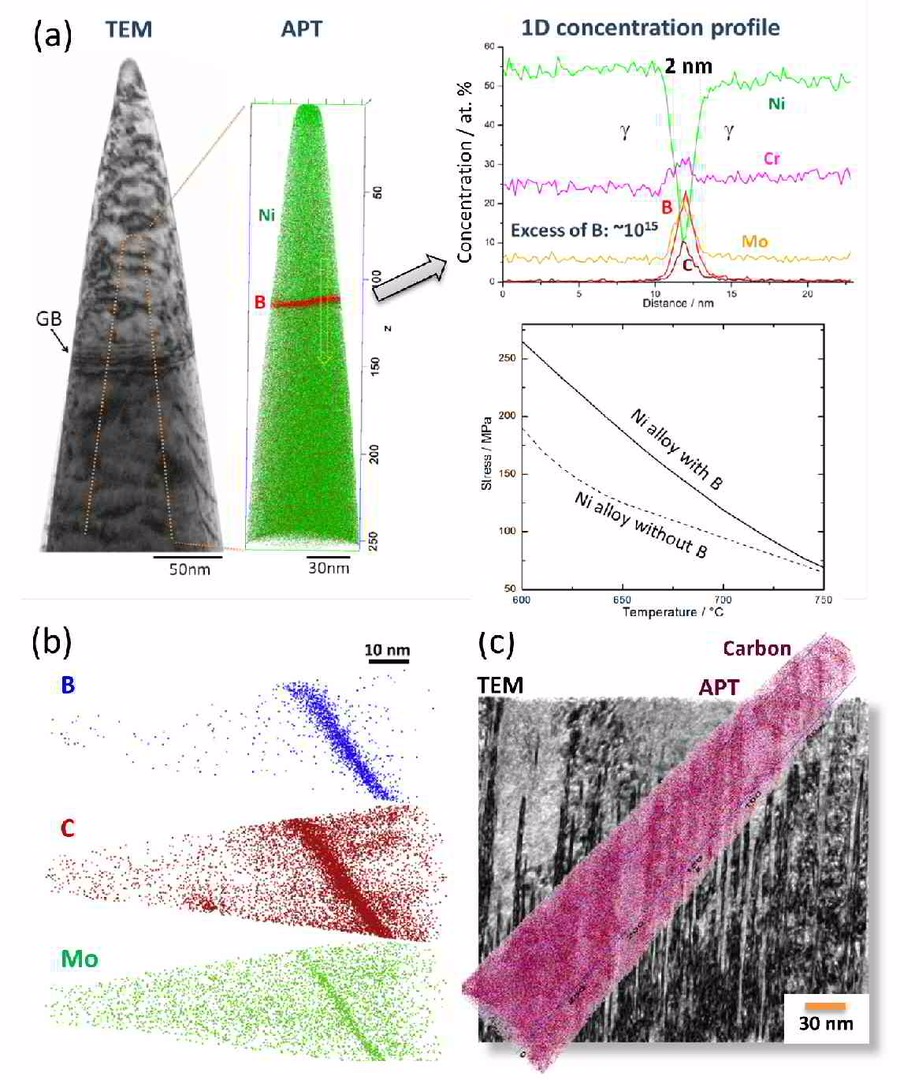
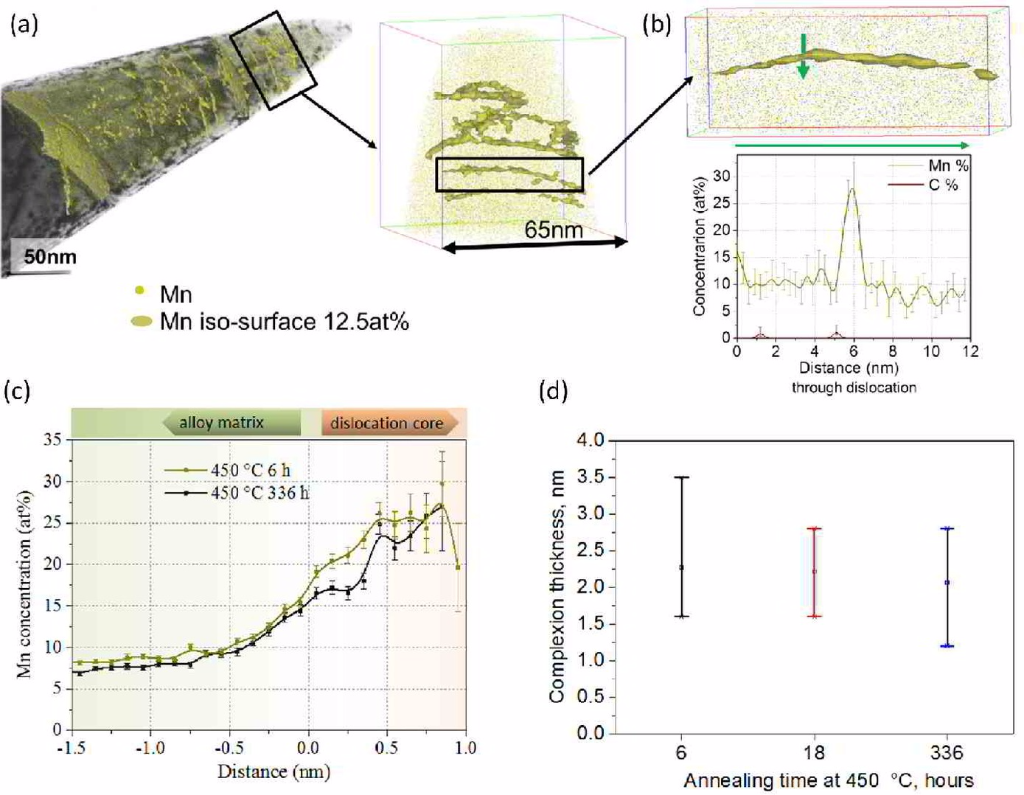
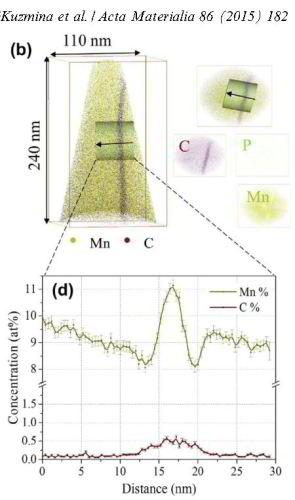
We study grain boundary embrittlement in a quenched and tempered Fe–Mn high-purity model martensite alloy using Charpy impact tests and grain boundary characterization by atom probe tomography. We observe that solute Mn directly embrittles martensite grain boundaries while reversion of martensite to austenite at high-angle grain boundaries cleans the interfaces from solute Mn by partitioning the Mn into the newly formed austenite, hence restoring impact toughness. Microalloying with B improves the impact toughness in the quenched state and delays temper embrittlement at 450 °C. Tempering at 600 °C for 1 min significantly improves the impact toughness and further tempering at lower temperature does not cause the embrittlement to return. At higher temperatures, regular austenite nucleation and growth takes place, whereas at lower temperature, Mn directly promotes its growth.
Acta Materialia 60 (2012) 1731-1740
Microstructural evolution of a Ni-based superalloy (617B) at 700 °C studied by electron microscopy and atom probe tomography
Acta Materialia 60 (2012) 1731 alloy 617[...]
PDF-Dokument [1.3 MB]
The demand for energy is set to double over the coming decades. In order to reduce CO2 emissions from fossil power plants there is an urgent need to improve energy conversion
efficiency. To achieve this goal, the next generation
of ultra-supercritical power plants must operate at service temperatures between 700 and 720 °C and steam pressures of up to 393 bar. However, conventional ferritic steels (P92) used
in current power plants cannot withstand such conditions, as they are designed for temperatures up to 600 °C and operating pressures up to 250 bar. Consequently, they are expected to
be replaced
by Ni-based superalloys which show longer creep rupture life and higher corrosion resistance.
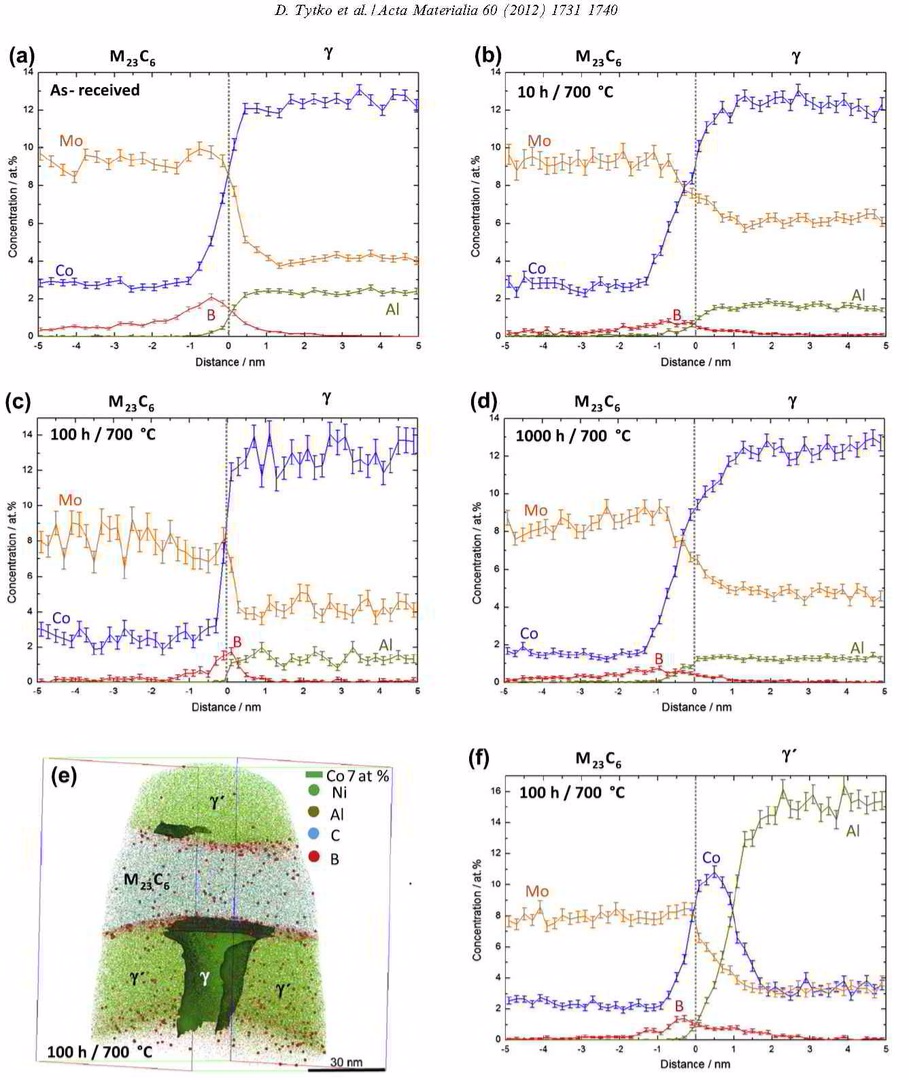
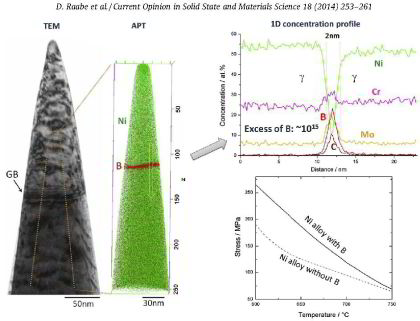
More specific, in this paper we report on the microstructural evolution of a polycrystalline Ni-based superalloy (Alloy 617B) for power plant applications at a
service temperature of 700 °C. The formation of secondary M23C6-carbides close to grain boundaries (GBs) and around primary Ti(C,N) particles is observed upon annealing at 700 °C, where gamma'
is found to nucleate heterogeneously at M23C6 carbides. Using atom probe tomography, elemental partitioning to the phases and composition profiles across phase and grain boundaries are
determined. Enrichments of B at gamma / M23C6 and gamma' / M23C6 interfaces as well as at grain boundaries are detected, while no B enrichment is found at gamma/gamma' interfaces. It
is suggested that segregation of B in conjunction with gamma' formation stabilizes a network of secondary M23C6 precipitates near GBs and thus increases the creep rupture life of Alloy
617B. Calculations of the equilibrium phase compositions by Thermo-Calc confirm the chemical compositions measured by atom probe tomography.
Acta Materialia 84 (2015) 110-123 atom p[...]
PDF-Dokument [2.4 MB]
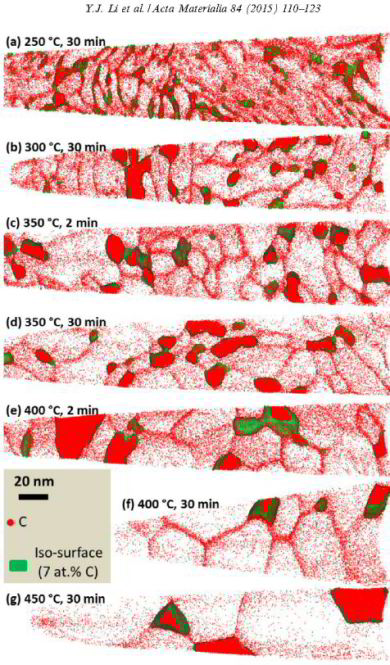
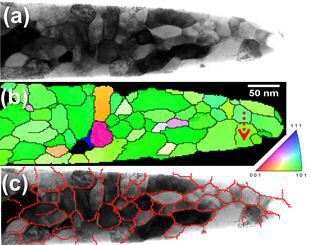
Carbon-supersaturated nanocrystalline hypereutectoid steels with a tensile strength of 6.35 GPa were produced from severely cold-drawn pearlite. The nanocrystalline material undergoes softening upon annealing at temperatures between 200 and 450 C. The ductility in terms of elongation to failure exhibits a non-monotonic dependence on temperature. Here, the microstructural mechanisms responsible for changes in the mechanical properties were studied using transmission electron microscopy (TEM), TEM-based automated scanning nanobeam diffraction and atom probe tomography (APT). TEM and APT investigations of the nanocrystalline hypereutectoid steel show subgrain coarsening upon annealing, which leads to strength reduction following a Hall–Petch law. APT analyzes of the Mn distribution near subgrain boundaries and in the cementite give strong evidence of capillary-driven subgrain coarsening occurring through subgrain boundary migration. The pronounced deterioration of ductility after annealing at temperatures above 350 C is attributed to the formation of cementite at subgrain boundaries. The overall segregation of carbon atoms at ferrite subgrain boundaries gives the nanocrystalline material excellent thermal stability upon annealing.
Acta Materialia 75 (2014) 147-155
Acta Materialia 75 (2014) 147 nanodiffus[...]
PDF-Dokument [787.4 KB]
A key requirement of modern steels – the combination of high strength and high deformability – can best be achieved by enabling a local adaptation of the microstructure during deformation. A local hardening is most efficiently obtained by a modification of the stacking sequence of atomic layers, resulting in the formation of twins or martensite. Combining ab initio calculations with in situ transmission electron microscopy, we show that the ability of a material to incorporate such stacking faults depends on its overall chemical composition and, importantly, the local composition near the defect, which is controlled by nanodiffusion. Specifically, the role of carbon for the stacking fault energy in high-Mn steels is investigated. Consequences for the long-term mechanical properties and the characterisation of these materials are discussed.
Phys Rev Lett. 2014 grain boundary segre[...]
PDF-Dokument [842.8 KB]
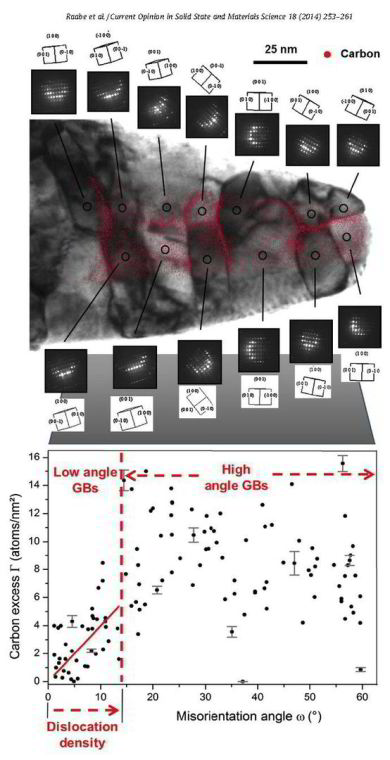
Grain boundary segregation leads to nanoscale chemical variations that can alter a material's performance by orders ofmagnitude (e.g., embrittlement). To understand this phenomenon, a large
number of grain boundaries
must be characterized in terms of both their five crystallographic interface parameters and their atomic-scale chemical composition. We demonstrate how this can be achieved using an approach
that combines the accuracy of structural characterization in transmission electron microscopy with the 3D chemical sensitivity of atom probe tomography.We find a linear trend between carbon
segregation and the misorientation angle ω for low-angle grain boundaries in ferrite, which indicates that ω is the most influential crystallographic parameter in this regime. However,
there are significant deviations from this linear trend indicating an additional strong
influence of other crystallographic parameters (grain boundary plane, rotation axis). For high-angle grain boundaries, no general trend between carbon excess and ω is observed; i.e., the grain
boundary plane and rotation axis have an even higher influence on the segregation behavior in this regime. Slight deviations from special grain boundary configurations are shown to lead to
unexpectedly high levels of segregation.
Current Opinion in Solid State and Materials Science 18 (2014) 253-261
Grain boundary segregation engineering in metallic alloys: A pathway to the design of interfaces
D. Raabe, M. Herbig, S. Sandlöbes, Y. Li, D. Tytko, M. Kuzmina, D. Ponge, P.-P. Choi
Current Opinion 2014 Grain Boundary Segr[...]
PDF-Dokument [1.7 MB]
Grain boundaries influence mechanical, functional, and kinetic properties of metallic alloys. They can be manipulated via solute decoration enabling changes in energy, mobility, structure,
and cohesion or even promoting local phase transformation. In the approach which we refer here to as ‘segregation engineering’ solute decoration is not regarded as an undesired phenomenon but is
instead utilized to manipulate specific grain boundary structures, compositions and properties that enable useful material behavior. The underlying thermodynamics follow the adsorption
isotherm. Hence, matrix-solute combinations suited for designing interfaces in metallic alloys can be identified by considering four main aspects, namely, the segregation coefficient of the
decorating element; its effects on interface cohesion, energy, structure
and mobility; its diffusion coefficient; and the free energies of competing bulk phases, precipitate phases or complexions. From a practical perspective, segregation engineering in alloys can be
usually realized by a modest diffusion heat treatment, hence, making it available in large scale manufacturing.
2014 scripta Mandal GB segregation 3D EB[...]
PDF-Dokument [972.8 KB]
Here we demonstrate a simplified nondestructive 3-D electron backscatter diffraction (EBSD) methodology that enables the measurement of all five degrees of freedom of grain boundaries (GBs) combined with segregation analysis using atom probe tomography (APT). The approach is based on two 2-D EBSD measurements on orthogonal surfaces at a sharp edge of the specimen followed by site-specific GB composition analysis using APT. An example of an asymmetric R9 boundary exhibiting GB segregation emphasizes the need for complete GB characterization in this context.
Adv Eng Mater 2014 Superalloy multiscale[...]
PDF-Dokument [2.8 MB]
In the present work, we show how conventional and advanced mechanical, chemical, and microstructural methods can be used to characterize cast single crystal Ni-base superalloy (SX)
plates across multiple length scales. Two types of microstructural heterogeneities are important, associated with the cast microstructure (dendrites (D) and interdendritic (ID) regions –
large scale heterogeneity) and with the well-known g/g0 microstructure (small scale heterogeneity). Using electron probe microanalysis (EPMA), we can show that elements such as Re, Co, and
Cr partition to the dendrites while ID regions contain more Al, Ta, and Ti. Analytical transmission electron microscopy and atom probe tomography (APT) show that Al, Ta, and Ti partition to
the g0 cubes while g channels show higher concentrations of Co, Cr, Re, andW.We can combine large scale (EPMA) and small-scale analytical methods (APT) to obtain reasonable estimates for g0
volume fractions in the dendrites and in the ID regions. The chemical and mechanical properties of the SX plates studied in the present work are homogeneous, when they are determined from
volumes with dimensions, which are significantly larger than the dendrite spacing. For the SX plates (140mm100mm20 mm) studied in the present work this holds for the average chemical
composition as well as for elastic behavior and local
creep properties. We highlight the potential of HRTEM and APT to contribute to a better understanding of the role of dislocations during coarsening of the g0 phase and the effect of
cooling rates after high temperature exposure on the microstructure.
2013-Acta-Nanocrystalline Fe–C alloys by[...]
PDF-Dokument [2.1 MB]
A series of nanocrystalline Fe–C alloys with different carbon concentrations (xtot) up to 19.4 at.% (4.90 wt.%) are prepared by ball milling. The microstructures of these alloys are
characterized by transmission electron microscopy and X-ray diffraction, and partitioning of carbon between grain boundaries and grain interiors is determined by atom probe tomography. It is
found that the segregation of carbon to grain boundaries of a-ferrite can significantly reduce its grain size to a few nanometers. When the grain boundaries of ferrite are saturated with
carbon, a metastable thermodynamic equilibrium between the matrix and the grain boundaries is approached, inducing a decreasing grain size with increasing xtot. Eventually the size reaches a
lower limit of about 6 nm in alloys with xtot > 6.19 at.%
(1.40 wt.%); a further increase in xtot leads to the precipitation of carbon as Fe3C. The observed presence of an amorphous structure in 19.4 at.% C (4.90 wt.%) alloy is ascribed to a
deformation-driven amorphization of Fe3C by severe plastic deformation. By measuring the temperature dependence of the grain size for an alloy with 1.77 at.% C additional evidence is provided
for a metastable equilibrium reached in the nanocrystalline alloy.
Raabe Acta Materialia 2013 Nanoscale-aus[...]
PDF-Dokument [6.0 MB]
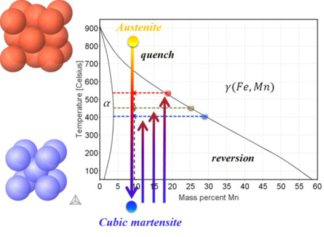
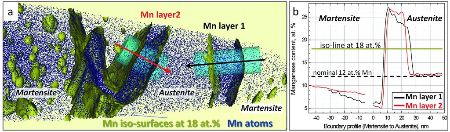
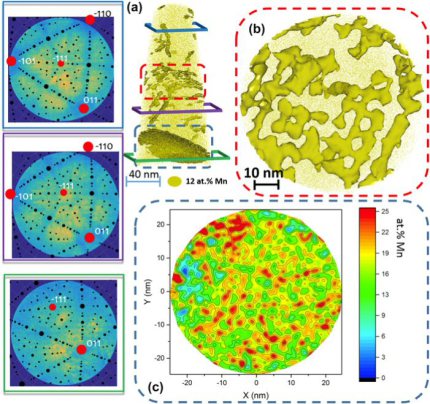
In an Fe–9 at.% Mn maraging alloy annealed at 450 C reversed allotriomorphic austenite nanolayers appear on former Mn decorated lath martensite boundaries. The austenite films are 5–15 nm
thick and form soft layers among the hard martensite crystals. We document the nanoscale segregation and associated martensite to austenite transformation mechanism using transmission
electron microscopy and atom probe tomography. The phenomena are discussed in terms of the adsorption isotherm (interface segregation) in conjunction with classical heterogeneous nucleation
theory (phase transformation) and a phase field model that predicts the kinetics
of phase transformation at segregation decorated grain boundaries. The analysis shows that strong interface segregation of austenite stabilizing elements (here Mn) and the release of elastic
stresses from the host martensite can generally promote phase transformation at martensite grain boundaries. The phenomenon enables the design of ductile and tough martensite.
Acta Mater 2014 GB segregation twin TWIP[...]
PDF-Dokument [665.4 KB]
We report on the characterization of grain boundary (GB) segregation in an Fe–28Mn–0.3C (wt.%) twinning-induced plasticity (TWIP) steel. After recrystallization of this steel for 24 h at 700
C, 50% general grain boundaries (GBs) and 35% R3 annealing twin boundaries were observed (others were high-order R and low-angle GBs). The segregation of B, C and P and traces of Si and Cu were
detected at the general GB by atom probe tomography (APT) and quantified using ladder diagrams. In the case of the R3 coherent annealing twin, it was necessary to first locate the position
of the boundary by density analysis of the atom probe data, then small amounts of B, Si and P segregation and, surprisingly, depletion of C were detected. The concentration of Mn was constant
across the interface for both boundary types. The depletion of C at the annealing twin is explained by a local change in the stacking sequence at the boundary, creating a local hexagonal
close-packed structure with low C solubility. This finding raises the question of whether segregation/depletion also occurs at R3 deformation twin boundaries in high-Mn TWIP steels.
Consequently, a previously published APT dataset of the Fe–22Mn–0.6C alloy system, containing a high density of deformation twins due to 30% tensile deformation at room temperature, was
reinvestigated using the same analysis routine as for the annealing twin. Although crystallographically identical to the
annealing twin, no evidence of segregation or depletion was found at the deformation twins, owing to the lack of mobility of solutes during twin
formation at room temperature.
2011_Acta_Materialia_APT_Steel-Fe-Mn.pdf
PDF-Dokument [2.3 MB]
Partitioning at phase boundaries of complex steels is important for their properties. We present atom probe tomography results across martensite/austenite interfaces in a
precipitation-hardened maraging-TRIP steel (12.2 Mn, 1.9 Ni, 0.6 Mo, 1.2 Ti, 0.3 Al; at.%). The system
reveals compositional changes at the phase boundaries: Mn and Ni are enriched while Ti, Al, Mo and Fe are depleted. More specific,
we observe up to 27 at.% Mn in a 20 nm layer at the phase boundary. This is explained by the large difference in diffusivity between martensite and austenite. The high diffusivity in martensite
leads to a Mn flux towards the retained austenite. The low diffusivity in the austenite does not allow accommodation of this flux. Consequently, the austenite grows with a Mn composition given
by local equilibrium. The interpretation is based on DICTRA and mixed-mode diffusion calculations (using a finite interface mobility).
Interfaces and defect composition at the near-atomic scale through atom probe tomography investigations
Here we review the use of Atom Probe Tomography in conjunction with electron microscopy-based probing techniques, to study the local composition of individual structural defects that differ
from their surrounding due to segregation.
These confined regions near or at defects exhibit their own partitioning behaviour and can be considered as separate confined phase states, sometimes also referred to as complexions.
Atom Probe Tomography is ideally suited to study segregation to crystalline defects such as grain boundraies and dislocations and the unique insights brought by the application of the technique
are critical to understand phenomena and processes at the nanoscale.
J. Mater. Res., 2018
Interfaces defect composition near-atomi[...]
PDF-Dokument [1.4 MB]